Polar opposites: Fine-tuning cytokinesis through SIN asymmetry
- PMID: 22786806
- PMCID: PMC3478943
- DOI: 10.1002/cm.21044
Polar opposites: Fine-tuning cytokinesis through SIN asymmetry
Abstract
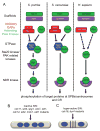
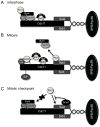
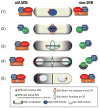
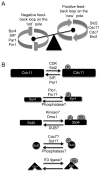
Mitotic exit and cell division must be spatially and temporally integrated to facilitate equal division of genetic material between daughter cells. In the fission yeast, Schizosaccharomyces pombe, a spindle pole body (SPB) localized signaling cascade termed the septation initiation network (SIN) couples mitotic exit with cytokinesis. The SIN is controlled at many levels to ensure that cytokinesis is executed once per cell cycle and only after cells segregate their DNA. An interesting facet of the SIN is that its activity is asymmetric on the two SPBs during anaphase; however, how and why the SIN is asymmetric has remained elusive. Many key factors controlling SIN asymmetry have now been identified, shedding light on the significance of SIN asymmetry in regulating cytokinesis. In this review, we highlight recent advances in our understanding of SIN regulation, with an emphasis on how SIN asymmetry is achieved and how this aspect of SIN regulation fine-tunes cytokinesis.
Copyright © 2012 Wiley Periodicals, Inc.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures
Similar articles
-
Nuc2p, a subunit of the anaphase-promoting complex, inhibits septation initiation network following cytokinesis in fission yeast.PLoS Genet. 2008 Jan;4(1):e17. doi: 10.1371/journal.pgen.0040017. PLoS Genet. 2008. PMID: 18225957 Free PMC article.
-
Relief of the Dma1-mediated checkpoint requires Dma1 autoubiquitination and dynamic localization.Mol Biol Cell. 2018 Sep 1;29(18):2176-2189. doi: 10.1091/mbc.E18-04-0261. Epub 2018 Jul 5. Mol Biol Cell. 2018. PMID: 29975113 Free PMC article.
-
SIN-inhibitory phosphatase complex promotes Cdc11p dephosphorylation and propagates SIN asymmetry in fission yeast.Curr Biol. 2011 Dec 6;21(23):1968-78. doi: 10.1016/j.cub.2011.10.051. Epub 2011 Nov 23. Curr Biol. 2011. PMID: 22119525 Free PMC article.
-
Pombe's thirteen - control of fission yeast cell division by the septation initiation network.J Cell Sci. 2015 Apr 15;128(8):1465-74. doi: 10.1242/jcs.094821. Epub 2015 Feb 17. J Cell Sci. 2015. PMID: 25690009 Review.
-
An overview of the fission yeast septation initiation network (SIN).Biochem Soc Trans. 2008 Jun;36(Pt 3):411-5. doi: 10.1042/BST0360411. Biochem Soc Trans. 2008. PMID: 18481970 Review.
Cited by
-
Localization of the ubiquitin ligase Dma1 to the fission yeast contractile ring is modulated by phosphorylation.FEBS Lett. 2021 Nov;595(22):2781-2792. doi: 10.1002/1873-3468.14211. Epub 2021 Nov 2. FEBS Lett. 2021. PMID: 34674264 Free PMC article.
-
Regulation of Yeast Cytokinesis by Calcium.J Fungi (Basel). 2025 Apr 2;11(4):278. doi: 10.3390/jof11040278. J Fungi (Basel). 2025. PMID: 40278099 Free PMC article. Review.
-
Comparative biology of cell division in the fission yeast clade.Curr Opin Microbiol. 2015 Dec;28:18-25. doi: 10.1016/j.mib.2015.07.011. Epub 2015 Aug 8. Curr Opin Microbiol. 2015. PMID: 26263485 Free PMC article. Review.
-
A novel checkpoint pathway controls actomyosin ring constriction trigger in fission yeast.Elife. 2020 Oct 26;9:e59333. doi: 10.7554/eLife.59333. Elife. 2020. PMID: 33103994 Free PMC article.
-
NDR Kinase Sid2 Drives Anillin-like Mid1 from the Membrane to Promote Cytokinesis and Medial Division Site Placement.Curr Biol. 2019 Mar 18;29(6):1055-1063.e2. doi: 10.1016/j.cub.2019.01.075. Epub 2019 Mar 7. Curr Biol. 2019. PMID: 30853434 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

