Pioglitazone, a PPARγ agonist, suppresses CYP19 transcription: evidence for involvement of 15-hydroxyprostaglandin dehydrogenase and BRCA1
- PMID: 22787115
- PMCID: PMC3694442
- DOI: 10.1158/1940-6207.CAPR-12-0201
Pioglitazone, a PPARγ agonist, suppresses CYP19 transcription: evidence for involvement of 15-hydroxyprostaglandin dehydrogenase and BRCA1
Retraction in
-
Retraction: Pioglitazone, a PPARγ Agonist, Suppresses CYP19 Transcription: Evidence for Involvement of 15-Hydroxyprostaglandin Dehydrogenase and BRCA1.Cancer Prev Res (Phila). 2022 Jun 2;15(6):411. doi: 10.1158/1940-6207.CAPR-22-0204. Cancer Prev Res (Phila). 2022. PMID: 35652223 Free PMC article. No abstract available.
Abstract
Estrogen synthesis is catalyzed by cytochrome P450 aromatase, which is encoded by the CYP19 gene. In obese postmenopausal women, increased aromatase activity in white adipose tissue is believed to contribute to hormone-dependent breast cancer. Prostaglandin E(2) (PGE(2)) stimulates the cAMP→protein kinase A (PKA) pathway leading to increased CYP19 transcription and elevated aromatase activity in inflamed white adipose tissue. 15-hydroxyprostaglandin dehydrogenase (15-PGDH) plays a major role in the catabolism of PGE(2). Here, we investigated the mechanism by which pioglitazone, a ligand of the nuclear receptor PPARγ suppressed aromatase expression. Treatment of human preadipocytes with pioglitazone suppressed Snail, a repressive transcription factor, resulting in elevated levels of 15-PGDH and reduced levels of PGE(2) in the culture medium. Pioglitazone also inhibited cAMP→PKA signaling leading to reduced interaction between phosphorylated cAMP responsive element-binding protein, p300, and CYP19 I.3/II promoter. BRCA1, a repressor of CYP19 transcription, was induced by pioglitazone. Consistent with these in vitro findings, treatment of mice with pioglitazone activated PPARγ, induced 15-PGDH and BRCA1 while suppressing aromatase levels in the mammary gland. Collectively, these results indicate that the activation of PPARγ induces BRCA1 and suppresses the PGE(2)→cAMP→PKA axis leading to reduced levels of aromatase. PPARγ agonists may have a role in reducing the risk of hormone-dependent breast cancer in obese postmenopausal women.
Conflict of interest statement
No potential conflicts of interest were disclosed.
Figures
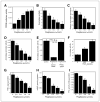
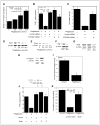


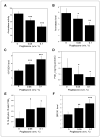

Comment in
-
Findings of Research Misconduct.Fed Regist. 2023 Sep 13;88(176):62800-62803. Fed Regist. 2023. PMID: 37736072 Free PMC article. No abstract available.
-
Findings of Research Misconduct.Fed Regist. 2023 Sep 13;88(176):62803-62807. Fed Regist. 2023. PMID: 37736073 Free PMC article. No abstract available.
References
-
- van Kruijsdijk RC, van der Wall E, Visseren FL. Obesity and cancer: the role of dysfunctional adipose tissue. Cancer Epidemiol Biomarkers Prev. 2009;18:2569–78. - PubMed
-
- Bulun SE, Lin Z, Imir G, Amin S, Demura M, Yilmaz B, et al. Regulation of aromatase expression in estrogen-responsive breast and uterine disease: from bench to treatment. Pharmacol Rev. 2005;57:359–83. - PubMed
-
- Lorincz AM, Sukumar S. Molecular links between obesity and breast cancer. Endocr Relat Cancer. 2006;13:279–92. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

