Integrating macrophages into organotypic co-cultures: a 3D in vitro model to study tumor-associated macrophages
- PMID: 22792213
- PMCID: PMC3391227
- DOI: 10.1371/journal.pone.0040058
Integrating macrophages into organotypic co-cultures: a 3D in vitro model to study tumor-associated macrophages
Abstract
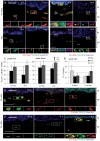
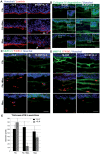
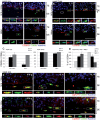
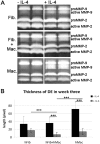
Tumor progression is controlled by signals from cellular and extra-cellular microenvironment including stromal cells and the extracellular matrix. Consequently, three-dimensional in vitro tumor models are essential to study the interaction of tumor cells with their microenvironment appropriately in a biologically relevant manner. We have previously used organotypic co-cultures to analyze the malignant growth of human squamous cell carcinoma (SCC) cell lines on a stromal equivalent in vitro. In this model, SCC cell lines are grown on a collagen-I gel containing fibroblasts. Since macrophages play a critical role in the progression of many tumor types, we now have expanded this model by integrating macrophages into the collagen gel of these organotypic tumor co-cultures. This model was established as a murine and a human system of skin SCCs. The effect of macrophages on tumor progression depends on their polarization. We demonstrate that macrophage polarization in organotypic co-cultures can be modulated towards and M1 or an M2 phenotype by adding recombinant IFN-γ and LPS or IL-4 respectively to the growth medium. IL-4 stimulation of macrophage-containing cultures resulted in enhanced tumor cell invasion evidenced by degradation of the basement membrane, enhanced collagenolytic activity and increased MMP-2 and MMP-9. Interestingly, extended co-culture with tumor cells for three weeks resulted in spontaneous M2 polarization of macrophages without IL-4 treatment. Thus, we demonstrate that macrophages can be successfully integrated into organotypic co-cultures of murine or human skin SCCs and that this model can be exploited to analyze macrophage activation towards a tumor supporting phenotype.
Conflict of interest statement
Figures


Similar articles
-
Cell type specific interleukin-6 induced responses in tumor keratinocytes and stromal fibroblasts are essential for invasive growth.Int J Cancer. 2014 Aug 1;135(3):551-62. doi: 10.1002/ijc.27951. Epub 2014 Mar 14. Int J Cancer. 2014. PMID: 23165423
-
Fibroblast-directed expression and localization of 92-kDa type IV collagenase along the tumor-stroma interface in an in vitro three-dimensional model of human squamous cell carcinoma.Mol Carcinog. 1997 Aug;19(4):258-66. doi: 10.1002/(sici)1098-2744(199708)19:4<258::aid-mc7>3.0.co;2-8. Mol Carcinog. 1997. PMID: 9290703
-
Oral squamous cell carcinoma-derived exosomes promote M2 subtype macrophage polarization mediated by exosome-enclosed miR-29a-3p.Am J Physiol Cell Physiol. 2019 May 1;316(5):C731-C740. doi: 10.1152/ajpcell.00366.2018. Epub 2019 Feb 27. Am J Physiol Cell Physiol. 2019. PMID: 30811223
-
3D Coculture between Cancer Cells and Macrophages: From Conception to Experimentation.ACS Biomater Sci Eng. 2024 Jan 8;10(1):313-325. doi: 10.1021/acsbiomaterials.3c01437. Epub 2023 Dec 18. ACS Biomater Sci Eng. 2024. PMID: 38110331 Review.
-
Special review series on 3D organotypic culture models: Introduction and historical perspective.In Vitro Cell Dev Biol Anim. 2021 Feb;57(2):95-103. doi: 10.1007/s11626-020-00500-2. Epub 2020 Nov 25. In Vitro Cell Dev Biol Anim. 2021. PMID: 33237402 Free PMC article. Review.
Cited by
-
A Reconstructed Human Melanoma-in-Skin Model to Study Immune Modulatory and Angiogenic Mechanisms Facilitating Initial Melanoma Growth and Invasion.Cancers (Basel). 2023 May 20;15(10):2849. doi: 10.3390/cancers15102849. Cancers (Basel). 2023. PMID: 37345186 Free PMC article.
-
3D Imaging Detection of HER2 Based in the Use of Novel Affibody-Quantum Dots Probes and Ratiometric Analysis.Transl Oncol. 2018 Jun;11(3):672-685. doi: 10.1016/j.tranon.2018.03.004. Epub 2018 Apr 5. Transl Oncol. 2018. PMID: 29627705 Free PMC article.
-
In vitro skin culture media influence the viability and inflammatory response of primary macrophages.Sci Rep. 2021 Mar 29;11(1):7070. doi: 10.1038/s41598-021-86486-7. Sci Rep. 2021. PMID: 33782484 Free PMC article.
-
Classic and new mediators for in vitro modelling of human macrophages.J Leukoc Biol. 2021 Mar;109(3):549-560. doi: 10.1002/JLB.1RU0620-018R. Epub 2020 Jun 27. J Leukoc Biol. 2021. PMID: 32592421 Free PMC article. Review.
-
An organotypic oral mucosal infection model to study host-pathogen interactions.J Tissue Eng. 2023 Oct 21;14:20417314231197310. doi: 10.1177/20417314231197310. eCollection 2023 Jan-Dec. J Tissue Eng. 2023. PMID: 37873034 Free PMC article.
References
-
- Mueller MM, Fusenig NE. Friends or foes - bipolar effects of the tumour stroma in cancer. Nat Rev Cancer. 2004;4:839–849. - PubMed
-
- Smola H, Stark HJ, Thiekotter G, Mirancea N, Krieg T, et al. Dynamics of basement membrane formation by keratinocyte-fibroblast interactions in organotypic skin culture. Exp Cell Res. 1998;239:399–410. - PubMed
-
- Hirschhaeuser F, Menne H, Dittfeld C, West J, Mueller-Klieser W, et al. Multicellular tumor spheroids: an underestimated tool is catching up again. J Biotechnol. 2010;148:3–15. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

