Perinatal maternal administration of Lactobacillus paracasei NCC 2461 prevents allergic inflammation in a mouse model of birch pollen allergy
- PMID: 22792257
- PMCID: PMC3391241
- DOI: 10.1371/journal.pone.0040271
Perinatal maternal administration of Lactobacillus paracasei NCC 2461 prevents allergic inflammation in a mouse model of birch pollen allergy
Abstract
Background: The hygiene hypothesis implies that microbial agents including probiotic bacteria may modulate foetal/neonatal immune programming and hence offer effective strategies for primary allergy prevention; however their mechanisms of action are poorly understood. We investigated whether oral administration of Lactobacillus paracasei NCC 2461 to mothers during gestation/lactation can protect against airway inflammation in offspring in a mouse model of birch pollen allergy, and examined the immune mechanisms involved.
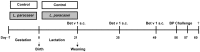
Methods: BALB/c mice were treated daily with L. paracasei in drinking water or drinking water alone in the last week of gestation and during lactation. Their offspring were sensitized with recombinant Bet v 1, followed by aerosol challenge with birch pollen extract.
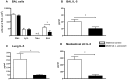
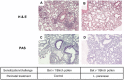
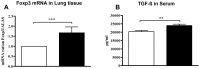
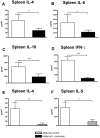
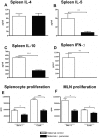
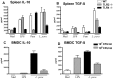
Results: Maternal exposure to L. paracasei prevented the development of airway inflammation in offspring, as demonstrated by attenuation of eosinophil influx in the lungs; reduction of IL-5 levels in bronchoalveolar lavage, and in lung and mediastinal lymph node cell cultures; and reduced peribronchial inflammatory infiltrate and mucus hypersecretion. While allergen-specific IgE and IgG antibody levels remained unchanged by the treatment, IL-4 and IL-5 production in spleen cell cultures were significantly reduced upon allergen stimulation in offspring of L. paracasei treated mice. Offspring of L. paracasei supplemented mothers had significantly reduced Bet v 1-specific as well as Concanavalin A-induced responses in spleen and mesenteric lymph node cell cultures, suggesting the modulation of both antigen-specific and mitogen-induced immune responses in offspring. These effects were associated with increased Foxp3 mRNA expression in the lungs and increased TGF-beta in serum.
Conclusion: Our data show that in a mouse model of birch pollen allergy, perinatal administration of L. paracasei NCC 2461 to pregnant/lactating mothers protects against the development of airway inflammation in offspring by activating regulatory pathways, likely through TLR2/4 signalling.
Conflict of interest statement
Figures
Similar articles
-
Prevention of birch pollen-related food allergy by mucosal treatment with multi-allergen-chimers in mice.PLoS One. 2012;7(6):e39409. doi: 10.1371/journal.pone.0039409. Epub 2012 Jun 29. PLoS One. 2012. PMID: 22768077 Free PMC article.
-
Oral exposure to Mal d 1 affects the immune response in patients with birch pollen allergy.J Allergy Clin Immunol. 2013 Jan;131(1):94-102. doi: 10.1016/j.jaci.2012.06.039. Epub 2012 Aug 24. J Allergy Clin Immunol. 2013. PMID: 22921871
-
Oocyst-Derived Extract of Toxoplasma Gondii Serves as Potent Immunomodulator in a Mouse Model of Birch Pollen Allergy.PLoS One. 2016 May 5;11(5):e0155081. doi: 10.1371/journal.pone.0155081. eCollection 2016. PLoS One. 2016. PMID: 27149118 Free PMC article.
-
Allergen immunotherapy for birch pollen-allergic patients: recent advances.Immunotherapy. 2016 May;8(5):555-67. doi: 10.2217/imt-2015-0027. Immunotherapy. 2016. PMID: 27140409 Review.
-
Birch pollen allergy in Europe.Allergy. 2019 Jul;74(7):1237-1248. doi: 10.1111/all.13758. Epub 2019 Apr 14. Allergy. 2019. PMID: 30829410 Review.
Cited by
-
Faecalibacterium prausnitzii Strain HTF-F and Its Extracellular Polymeric Matrix Attenuate Clinical Parameters in DSS-Induced Colitis.PLoS One. 2015 Apr 24;10(4):e0123013. doi: 10.1371/journal.pone.0123013. eCollection 2015. PLoS One. 2015. PMID: 25910186 Free PMC article.
-
House dust mite allergy: environment evaluation and disease prevention.Asia Pac Allergy. 2014 Oct;4(4):241-52. doi: 10.5415/apallergy.2014.4.4.241. Epub 2014 Oct 29. Asia Pac Allergy. 2014. PMID: 25379484 Free PMC article. Review.
-
Bifidobacterium longum CCM 7952 Promotes Epithelial Barrier Function and Prevents Acute DSS-Induced Colitis in Strictly Strain-Specific Manner.PLoS One. 2015 Jul 28;10(7):e0134050. doi: 10.1371/journal.pone.0134050. eCollection 2015. PLoS One. 2015. PMID: 26218526 Free PMC article.
-
Polysaccharides L900/2 and L900/3 isolated from Lactobacillus rhamnosus LOCK 0900 modulate allergic sensitization to ovalbumin in a mouse model.Microb Biotechnol. 2017 May;10(3):586-593. doi: 10.1111/1751-7915.12606. Epub 2017 Feb 6. Microb Biotechnol. 2017. PMID: 28165193 Free PMC article.
-
Colonization of germ-free mice with a mixture of three lactobacillus strains enhances the integrity of gut mucosa and ameliorates allergic sensitization.Cell Mol Immunol. 2016 Mar;13(2):251-62. doi: 10.1038/cmi.2015.09. Epub 2015 Mar 23. Cell Mol Immunol. 2016. PMID: 25942514 Free PMC article.
References
-
- Moffatt MF, Kabesch M, Liang L, Dixon AL, Strachan D, et al. Genetic variants regulating ORMDL3 expression contribute to the risk of childhood asthma. Nature. 2007;448:470–473. - PubMed
-
- Vercelli D. Discovering susceptibility genes for asthma and allergy. Nat Rev Immunol. 2008;8:169–182. - PubMed
-
- Savino F, Cresi F, Pautasso S, Palumeri E, Tullio V, et al. Intestinal microflora in breastfed colicky and non-colicky infants. Acta Paediatr. 2004;93:825–829. - PubMed
-
- Savino F, Roana J, Mandras N, Tarasco V, Locatelli E, et al. Faecal microbiota in breast-fed infants after antibiotic therapy. Acta Paediatr. 2011;100:75–78. - PubMed
-
- Bjorksten B, Naaber P, Sepp E, Mikelsaar M. The intestinal microflora in allergic Estonian and Swedish 2-year-old children. Clin Exp Allergy. 1999;29:342–346. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

