Characteristic features of kynurenine aminotransferase allosterically regulated by (alpha)-ketoglutarate in cooperation with kynurenine
- PMID: 22792273
- PMCID: PMC3391261
- DOI: 10.1371/journal.pone.0040307
Characteristic features of kynurenine aminotransferase allosterically regulated by (alpha)-ketoglutarate in cooperation with kynurenine
Abstract
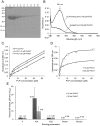
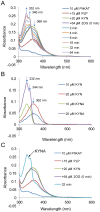
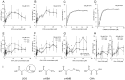
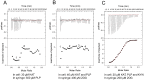
Kynurenine aminotransferase from Pyrococcus horikoshii OT3 (PhKAT), which is a homodimeric protein, catalyzes the conversion of kynurenine (KYN) to kynurenic acid (KYNA). We analyzed the transaminase reaction mechanisms of this protein with pyridoxal-5'-phosphate (PLP), KYN and α-ketoglutaric acid (2OG) or oxaloacetic acid (OXA). 2OG significantly inhibited KAT activities in kinetic analyses, suggesting that a KYNA biosynthesis is allosterically regulated by 2OG. Its inhibitions evidently were unlocked by KYN. 2OG and KYN functioned as an inhibitor and activator in response to changes in the concentrations of KYN and 2OG, respectively. The affinities of one subunit for PLP or 2OG were different from that of the other subunit, as confirmed by spectrophotometry and isothermal titration calorimetry, suggesting that the difference of affinities between subunits might play a role in regulations of the KAT reaction. Moreover, we identified two active and allosteric sites in the crystal structure of PhKAT-2OG complexes. The crystal structure of PhKAT in complex with four 2OGs demonstrates that two 2OGs in allosteric sites are effector molecules which inhibit the KYNA productions. Thus, the combined data lead to the conclusion that PhKAT probably is regulated by allosteric control machineries, with 2OG as the allosteric inhibitor.
Conflict of interest statement
Figures



Similar articles
-
Structural and mechanistic insights into the kynurenine aminotransferase-mediated excretion of kynurenic acid.J Struct Biol. 2014 Mar;185(3):257-66. doi: 10.1016/j.jsb.2014.01.009. Epub 2014 Jan 25. J Struct Biol. 2014. PMID: 24473062
-
Quantum mechanics/molecular mechanics (QM/MM) modeling of the irreversible transamination of L-kynurenine to kynurenic acid: the round dance of kynurenine aminotransferase II.Biochim Biophys Acta. 2009 Dec;1794(12):1802-12. doi: 10.1016/j.bbapap.2009.08.016. Epub 2009 Aug 26. Biochim Biophys Acta. 2009. PMID: 19715778
-
Structure of the PLP-Form of the Human Kynurenine Aminotransferase II in a Novel Spacegroup at 1.83 Å Resolution.Int J Mol Sci. 2016 Mar 25;17(4):446. doi: 10.3390/ijms17040446. Int J Mol Sci. 2016. PMID: 27023527 Free PMC article.
-
Kynurenine Aminotransferase Isozyme Inhibitors: A Review.Int J Mol Sci. 2016 Jun 15;17(6):946. doi: 10.3390/ijms17060946. Int J Mol Sci. 2016. PMID: 27314340 Free PMC article. Review.
-
The role of glutamine transaminase K (GTK) in sulfur and alpha-keto acid metabolism in the brain, and in the possible bioactivation of neurotoxicants.Neurochem Int. 2004 Jun;44(8):557-77. doi: 10.1016/j.neuint.2003.12.002. Neurochem Int. 2004. PMID: 15016471 Review.
Cited by
-
Mitochondrial Impairment: A Common Motif in Neuropsychiatric Presentation? The Link to the Tryptophan-Kynurenine Metabolic System.Cells. 2022 Aug 21;11(16):2607. doi: 10.3390/cells11162607. Cells. 2022. PMID: 36010683 Free PMC article. Review.
-
Biochemical Characterization of Aspergillus fumigatus AroH, a Putative Aromatic Amino Acid Aminotransferase.Front Mol Biosci. 2018 Nov 28;5:104. doi: 10.3389/fmolb.2018.00104. eCollection 2018. Front Mol Biosci. 2018. PMID: 30547035 Free PMC article.
-
Biochemical and genetic examination of two aminotransferases from the hyperthermophilic archaeon Thermococcus kodakarensis.Front Microbiol. 2023 Feb 20;14:1126218. doi: 10.3389/fmicb.2023.1126218. eCollection 2023. Front Microbiol. 2023. PMID: 36891395 Free PMC article.
-
Tryptophan catabolism in Pseudomonas aeruginosa and potential for inter-kingdom relationship.BMC Microbiol. 2016 Jul 8;16(1):137. doi: 10.1186/s12866-016-0756-x. BMC Microbiol. 2016. PMID: 27392067 Free PMC article.
-
Metabolomic analysis of rumen fluid in Tan sheep reveals sex-specific key metabolites and pathways associated with residual feed intake.Sci Rep. 2025 Jul 1;15(1):22115. doi: 10.1038/s41598-025-06182-8. Sci Rep. 2025. PMID: 40594387 Free PMC article.
References
-
- Tobes MC, Mason M. Alpha-Aminoadipate aminotransferase and kynurenine aminotransferase. Purification, characterization, and further evidence for identity. J Biol Chem. 1977;252:4591–4599. - PubMed
-
- Hartai Z, Juhasz A, Rimanoczy A, Janaky T, Donko T, et al. Decreased serum and red blood cell kynurenic acid levels in Alzheimer’s disease. Neurochem Int. 2007;50:308–313. - PubMed
-
- Erhardt S, Schwieler L, Engberg G. Kynurenic acid and schizophrenia. Adv Exp Med Biol. 2003;527:155–165. - PubMed
-
- Rossi F, Schwarcz R, Rizzi M. Curiosity to kill the KAT (kynurenine aminotransferase): structural insights into brain kynurenic acid synthesis. Curr Opin Struct Biol. 2008;18:748–755. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Research Materials

