Memantine inhibits α3β2-nAChRs-mediated nitrergic neurogenic vasodilation in porcine basilar arteries
- PMID: 22792283
- PMCID: PMC3390354
- DOI: 10.1371/journal.pone.0040326
Memantine inhibits α3β2-nAChRs-mediated nitrergic neurogenic vasodilation in porcine basilar arteries
Abstract
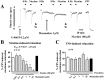
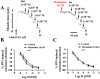
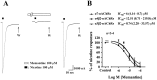
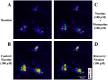
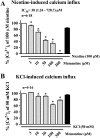
Memantine, an NMDA receptor antagonist used for treatment of Alzheimer's disease (AD), is known to block the nicotinic acetylcholine receptors (nAChRs) in the central nervous system (CNS). In the present study, we examined by wire myography if memantine inhibited α3β2-nAChRs located on cerebral perivascular sympathetic nerve terminals originating in the superior cervical ganglion (SCG), thus, leading to inhibition of nicotine-induced nitrergic neurogenic dilation of isolated porcine basilar arteries. Memantine concentration-dependently blocked nicotine-induced neurogenic dilation of endothelium-denuded basilar arteries without affecting that induced by transmural nerve stimulation, sodium nitroprusside, or isoproterenol. Furthermore, memantine significantly inhibited nicotine-elicited inward currents in Xenopous oocytes expressing α3β2-, α7- or α4β2-nAChR, and nicotine-induced calcium influx in cultured rat SCG neurons. These results suggest that memantine is a non-specific antagonist for nAChR. By directly inhibiting α3β2-nAChRs located on the sympathetic nerve terminals, memantine blocks nicotine-induced neurogenic vasodilation of the porcine basilar arteries. This effect of memantine is expected to reduce the blood supply to the brain stem and possibly other brain regions, thus, decreasing its clinical efficacy in the treatment of Alzheimer's disease.
Conflict of interest statement
Figures
Similar articles
-
L-type calcium channels in sympathetic α3β2-nAChR-mediated cerebral nitrergic neurogenic vasodilation.Acta Physiol (Oxf). 2014 Aug;211(4):544-58. doi: 10.1111/apha.12315. Epub 2014 Jun 9. Acta Physiol (Oxf). 2014. PMID: 24825168
-
Inhibition by ketamine and amphetamine analogs of the neurogenic nitrergic vasodilations in porcine basilar arteries.Toxicol Appl Pharmacol. 2016 Aug 15;305:75-82. doi: 10.1016/j.taap.2016.05.020. Epub 2016 Jun 1. Toxicol Appl Pharmacol. 2016. PMID: 27260673
-
Pb2+ inhibition of sympathetic alpha 7-nicotinic acetylcholine receptor-mediated nitrergic neurogenic dilation in porcine basilar arteries.J Pharmacol Exp Ther. 2003 Jun;305(3):1124-31. doi: 10.1124/jpet.102.046854. Epub 2003 Mar 6. J Pharmacol Exp Ther. 2003. PMID: 12626657
-
Sympathetic modulation of nitrergic neurogenic vasodilation in cerebral arteries.Jpn J Pharmacol. 2002 Jan;88(1):26-31. doi: 10.1254/jjp.88.26. Jpn J Pharmacol. 2002. PMID: 11859855 Review.
-
Axo-axonal interaction in autonomic regulation of the cerebral circulation.Acta Physiol (Oxf). 2011 Sep;203(1):25-35. doi: 10.1111/j.1748-1716.2010.02231.x. Acta Physiol (Oxf). 2011. PMID: 21159131 Review.
Cited by
-
Randomized Placebo-Controlled Trial of Memantine for Smoking Cessation (CCCWFU 99311).Cancer Control. 2025 Jan-Dec;32:10732748251336416. doi: 10.1177/10732748251336416. Epub 2025 May 8. Cancer Control. 2025. PMID: 40340434 Free PMC article. Clinical Trial.
-
Therapeutics of Neurotransmitters in Alzheimer's Disease.J Alzheimers Dis. 2017;57(4):1049-1069. doi: 10.3233/JAD-161118. J Alzheimers Dis. 2017. PMID: 28211810 Free PMC article. Review.
-
Indoor Secondary Pollutants Cannot Be Ignored: Third-Hand Smoke.Toxics. 2022 Jun 30;10(7):363. doi: 10.3390/toxics10070363. Toxics. 2022. PMID: 35878269 Free PMC article. Review.
-
Acute and chronic glutamate NMDA antagonist treatment attenuates dopamine D1 antagonist-induced reduction of nicotine self-administration in female rats.Pharmacol Biochem Behav. 2024 Jan;234:173678. doi: 10.1016/j.pbb.2023.173678. Epub 2023 Nov 16. Pharmacol Biochem Behav. 2024. PMID: 37979731 Free PMC article.
-
Activation of Neuropeptide Y2 Receptor Can Inhibit Global Cerebral Ischemia-Induced Brain Injury.Neuromolecular Med. 2022 Jun;24(2):97-112. doi: 10.1007/s12017-021-08665-z. Epub 2021 May 21. Neuromolecular Med. 2022. PMID: 34019239 Free PMC article.
References
-
- Castellani RJ, Lee HG, Zhu X, Nunomura A, Perry G, et al. Neuropathology of Alzheimer disease: pathognomonic but not pathogenic. Acta Neuropathol. 2006;111:503–509. - PubMed
-
- Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science. 2002;297:353–356. - PubMed
-
- Nussbaum RL, Ellis CE. Alzheimer’s disease and Parkinson’s disease. N Engl J Med. 2003;348:1356–1364. - PubMed
-
- Rogawski MA. Low affinity channel blocking (uncompetitive) NMDA receptor antagonists as therapeutic agents–toward an understanding of their favorable tolerability. Amino Acids. 2000;19:133–149. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

