Loss of the integrin-activating transmembrane protein Fam38A (Piezo1) promotes a switch to a reduced integrin-dependent mode of cell migration
- PMID: 22792288
- PMCID: PMC3390408
- DOI: 10.1371/journal.pone.0040346
Loss of the integrin-activating transmembrane protein Fam38A (Piezo1) promotes a switch to a reduced integrin-dependent mode of cell migration
Abstract
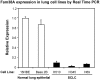
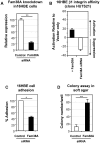
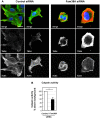
Lung cancer is one of the most common fatal diseases in the developed world. The disease is rarely cured by currently available therapies, with an overall survival rate of ∼10%. Characterizing novel proteins that offer crucial insights into the processes of lung tumour invasion and metastasis may therefore provide much-needed prognostic markers, and influence therapeutic strategies. Aberrant function of the integrin family of heterodimeric cell surface receptors is a common theme in cancer--investigation into novel integrin activity regulators may offer crucial insights into the processes of tumour invasion and metastasis and may reveal insights into potential therapeutic targets. We previously described that depletion of the novel multi-transmembrane domain protein Fam38A, located at the endoplasmic reticulum (ER), inactivates endogenous beta1 integrin affinity, reducing cell adhesion. We now show that depletion of Fam38A, also now known as Piezo1, causes anchorage independence and a switch to a reduced integrin-dependent mode of cell migration/invasion, a novel phenotype for this integrin-regulating protein. Normal lung epithelial cells show increased rates of migration by 2D time-lapse microscopy and increased capacity to invade into matrigel, despite having decreased integrin affinity. We confirm greatly depleted Fam38A expression in small cell lung cancer (SCLC) lines where a form of reduced integrin-dependent migration, i.e. amoeboid migration, is a known phenotype. We propose that loss of Fam38A expression may cause increased cell migration and metastasis in lung tumours.
Conflict of interest statement
Figures

Similar articles
-
Integrin activation by Fam38A uses a novel mechanism of R-Ras targeting to the endoplasmic reticulum.J Cell Sci. 2010 Jan 1;123(Pt 1):51-61. doi: 10.1242/jcs.056424. J Cell Sci. 2010. PMID: 20016066 Free PMC article.
-
Calpain 2 and Src dependence distinguishes mesenchymal and amoeboid modes of tumour cell invasion: a link to integrin function.Oncogene. 2006 Sep 21;25(42):5726-40. doi: 10.1038/sj.onc.1209582. Epub 2006 May 1. Oncogene. 2006. PMID: 16652152
-
Integrin α9β1 promotes malignant tumor growth and metastasis by potentiating epithelial-mesenchymal transition.Oncogene. 2013 Jan 10;32(2):141-50. doi: 10.1038/onc.2012.41. Epub 2012 Feb 27. Oncogene. 2013. PMID: 22370635 Free PMC article.
-
Activating Invasion and Metastasis in Small Cell Lung Cancer: Role of the Tumour Immune Microenvironment and Mechanisms of Vasculogenesis, Epithelial-Mesenchymal Transition, Cell Migration, and Organ Tropism.Cancer Rep (Hoboken). 2024 Oct;7(10):e70018. doi: 10.1002/cnr2.70018. Cancer Rep (Hoboken). 2024. PMID: 39376011 Free PMC article. Review.
-
Integrin receptors in primary lung cancer.Exp Oncol. 2004 Jun;26(2):106-10. Exp Oncol. 2004. PMID: 15273661 Review.
Cited by
-
Piezo1 Is Required for Myoblast Migration and Involves Polarized Clustering in Association with Cholesterol and GM1 Ganglioside.Cells. 2023 Dec 7;12(24):2784. doi: 10.3390/cells12242784. Cells. 2023. PMID: 38132106 Free PMC article.
-
Aberrant overexpression of myosin 1b in glioblastoma promotes angiogenesis via VEGF-myc-myosin 1b-Piezo1 axis.J Biol Chem. 2024 Nov;300(11):107807. doi: 10.1016/j.jbc.2024.107807. Epub 2024 Sep 20. J Biol Chem. 2024. PMID: 39307302 Free PMC article.
-
Mechanosensitive Ion Channels and Their Role in Cancer Cells.Membranes (Basel). 2023 Jan 29;13(2):167. doi: 10.3390/membranes13020167. Membranes (Basel). 2023. PMID: 36837670 Free PMC article. Review.
-
Fine-Tuning of Piezo1 Expression and Activity Ensures Efficient Myoblast Fusion during Skeletal Myogenesis.Cells. 2022 Jan 24;11(3):393. doi: 10.3390/cells11030393. Cells. 2022. PMID: 35159201 Free PMC article.
-
Inside Out Integrin Activation Mediated by PIEZO1 Signaling in Erythroblasts.Front Physiol. 2020 Jul 31;11:958. doi: 10.3389/fphys.2020.00958. eCollection 2020. Front Physiol. 2020. PMID: 32848880 Free PMC article.
References
-
- Levi F, Lucchini F, Negri E, Boyle P, La Vecchia C. Cancer mortality in Europe, 1995–1999, and an overview of trends since 1960. Int J Cancer. 2004;110:155–169. - PubMed
-
- Hood JD, Cheresh DA. Role of integrins in cell invasion and migration. Nat Rev Cancer. 2002;2:91–100. - PubMed
-
- Danen EH, Yamada KM. Fibronectin, integrins, and growth control. J Cell Physiol. 2001;189:1–13. - PubMed
-
- Plow EF, Haas TA, Zhang L, Loftus J, Smith JW. Ligand binding to integrins. J Biol Chem. 2000;275:21785–21788. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

