Staphylococcus aureus protein A plays a critical role in mediating bone destruction and bone loss in osteomyelitis
- PMID: 22792377
- PMCID: PMC3394727
- DOI: 10.1371/journal.pone.0040586
Staphylococcus aureus protein A plays a critical role in mediating bone destruction and bone loss in osteomyelitis
Abstract
Staphylococcus aureus is the most frequent causative organism of osteomyelitis. It is characterised by widespread bone loss and bone destruction. Previously we demonstrated that S. aureus protein A (SpA) is capable of binding to tumour necrosis factor receptor-1 expressed on pre-osteoblastic cells, which results in signal generation that leads to cell apoptosis resulting in bone loss. In the current report we demonstrate that upon S. aureus binding to osteoblasts it also inhibits de novo bone formation by preventing expression of key markers of osteoblast growth and division such as alkaline phosphatase, collagen type I, osteocalcin, osteopontin and osteocalcin. In addition, S. aureus induces secretion of soluble RANKL from osteoblasts which in turn recruits and activates the bone resorbing cells, osteoclasts. A strain of S. aureus defective in SpA failed to affect osteoblast growth or proliferation and most importantly failed to recruit or activate osteoclasts. These results suggest that S. aureus SpA binding to osteoblasts provides multiple coordinated signals that accounts for bone loss and bone destruction seen in osteomyelitis cases. A better understanding of the mechanisms through which S. aureus leads to bone infection may improve treatment or lead to the development of better therapeutic agents to treat this notoriously difficult disease.
Conflict of interest statement
Figures
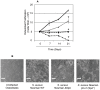
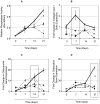
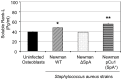
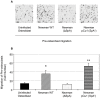
Similar articles
-
Staphylococcus aureus protein A binds to osteoblasts and triggers signals that weaken bone in osteomyelitis.PLoS One. 2011 Apr 15;6(4):e18748. doi: 10.1371/journal.pone.0018748. PLoS One. 2011. PMID: 21525984 Free PMC article.
-
Staphylococcus aureus protein A binding to osteoblast tumour necrosis factor receptor 1 results in activation of nuclear factor kappa B and release of interleukin-6 in bone infection.Microbiology (Reading). 2013 Jan;159(Pt 1):147-154. doi: 10.1099/mic.0.063016-0. Epub 2012 Nov 15. Microbiology (Reading). 2013. PMID: 23154968
-
Staphylococcus aureus biofilms decrease osteoblast viability, inhibits osteogenic differentiation, and increases bone resorption in vitro.BMC Musculoskelet Disord. 2013 Jun 14;14:187. doi: 10.1186/1471-2474-14-187. BMC Musculoskelet Disord. 2013. PMID: 23767824 Free PMC article.
-
Staphylococcus aureus vs. Osteoblast: Relationship and Consequences in Osteomyelitis.Front Cell Infect Microbiol. 2015 Nov 26;5:85. doi: 10.3389/fcimb.2015.00085. eCollection 2015. Front Cell Infect Microbiol. 2015. PMID: 26636047 Free PMC article. Review.
-
Intracellular Staphylococcus aureus in osteoblasts and osteocytes and its impact on bone homeostasis during osteomyelitis.Bone. 2025 Sep;198:117536. doi: 10.1016/j.bone.2025.117536. Epub 2025 May 18. Bone. 2025. PMID: 40393553 Review.
Cited by
-
In Silico Genome-Scale Analysis of Molecular Mechanisms Contributing to the Development of a Persistent Infection with Methicillin-Resistant Staphylococcus aureus (MRSA) ST239.Int J Mol Sci. 2022 Dec 16;23(24):16086. doi: 10.3390/ijms232416086. Int J Mol Sci. 2022. PMID: 36555727 Free PMC article.
-
Exploiting Correlations between Protein Abundance and the Functional Status of saeRS and sarA To Identify Virulence Factors of Potential Importance in the Pathogenesis of Staphylococcus aureus Osteomyelitis.ACS Infect Dis. 2020 Feb 14;6(2):237-249. doi: 10.1021/acsinfecdis.9b00291. Epub 2019 Nov 26. ACS Infect Dis. 2020. PMID: 31722523 Free PMC article.
-
Metabolic activity, urease production, antibiotic resistance and virulence in dual species biofilms of Staphylococcus epidermidis and Staphylococcus aureus.PLoS One. 2017 Mar 6;12(3):e0172700. doi: 10.1371/journal.pone.0172700. eCollection 2017. PLoS One. 2017. PMID: 28263995 Free PMC article.
-
Staphylococcus aureus peptidoglycan promotes osteoclastogenesis via TLR2-mediated activation of the NF-κB/NFATc1 signaling pathway.Am J Transl Res. 2017 Nov 15;9(11):5022-5030. eCollection 2017. Am J Transl Res. 2017. PMID: 29218100 Free PMC article.
-
The Increased Accumulation of Staphylococcus aureus Virulence Factors Is Maximized in a purR Mutant by the Increased Production of SarA and Decreased Production of Extracellular Proteases.Infect Immun. 2021 Mar 17;89(4):e00718-20. doi: 10.1128/IAI.00718-20. Print 2021 Mar 17. Infect Immun. 2021. PMID: 33468580 Free PMC article.
References
-
- Whyte MP. Hypophosphatasia and the role of alkaline phosphatase in skeletal mineralization. Endocr Rev. 1994;15:439–461. - PubMed
-
- Brodsky B, Persikov AV. Molecular structure of the collagen triple helix. Adv Protein Chem. 2005;70:301–339. - PubMed
-
- Roach HI. Why does bone matrix contain non-collagenous proteins? The possible roles of osteocalcin, osteonectin, osteopontin and bone sialoprotein in bone mineralisation and resorption. Cell Biol Int. 1994;18:617–628. - PubMed
-
- Roodman GD. Advances in bone biology: the osteoclast. Endocr Rev. 1996;17:308–332. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

