Interstage mortality after the Norwood procedure: Results of the multicenter Single Ventricle Reconstruction trial
- PMID: 22795436
- PMCID: PMC3985484
- DOI: 10.1016/j.jtcvs.2012.05.020
Interstage mortality after the Norwood procedure: Results of the multicenter Single Ventricle Reconstruction trial
Abstract
Objective: For infants with single ventricle malformations undergoing staged repair, interstage mortality is reported at 2% to 20%. The Single Ventricle Reconstruction trial randomized subjects with a single morphologic right ventricle undergoing a Norwood procedure to a modified Blalock-Taussig shunt (MBTS) or a right ventricle-to-pulmonary artery shunt (RVPAS). The aim of this analysis was to explore the associations of interstage mortality and shunt type, and demographic, anatomic, and perioperative factors.
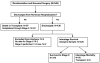
Methods: Participants in the Single Ventricle Reconstruction trial who survived to discharge after the Norwood procedure were included (n = 426). Interstage mortality was defined as death postdischarge after the Norwood procedure and before the stage II procedure. Univariate analysis and multivariable logistic regression were performed adjusting for site.
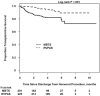
Results: Overall interstage mortality was 50 of 426 (12%)-13 of 225 (6%) for RVPAS and 37 of 201 (18%) for MBTS (odds ratio [OR] for MBTS, 3.4; P < .001). When moderate to severe postoperative atrioventricular valve regurgitation (AVVR) was present, interstage mortality was similar between shunt types. Interstage mortality was independently associated with gestational age less than 37 weeks (OR, 3.9; P = .008), Hispanic ethnicity (OR, 2.6; P = .04), aortic atresia/mitral atresia (OR, 2.3; P = .03), greater number of post-Norwood complications (OR, 1.2; P = .006), census block poverty level (P = .003), and MBTS in subjects with no or mild postoperative AVVR (OR, 9.7; P < .001).
Conclusions: Interstage mortality remains high at 12% and is increased with the MBTS compared with the RVPAS if postoperative AVVR is absent or mild. Preterm delivery, anatomic, and socioeconomic factors are also important. Avoiding preterm delivery when possible and close surveillance after Norwood hospitalization for infants with identified risk factors may reduce interstage mortality.
Trial registration: ClinicalTrials.gov NCT00115934.
Copyright © 2012 The American Association for Thoracic Surgery. Published by Mosby, Inc. All rights reserved.
Figures
References
-
- Simsic JM, Bradley SM, Stroud MR, Atz AM. Risk factors for interstage death after the Norwood procedure. Pediatr Cardiol. 2005;26:400–3. - PubMed
-
- Ghanayem NS, Tweddell JS, Hoffman GM, Mussatto K, Jaquiss RD. Optimal timing of the second stage of palliation for hypoplastic left heart syndrome facilitated through home monitoring, and the results of early cavopulmonary anastomosis. Cardiol Young. 2006;16(Suppl I):60–5. - PubMed
-
- Hehir DA, Dominguez TE, Ballweg JA, Ravishankar C, Marino BS, Bird GL, et al. Risk factors for interstage death after stage 1 reconstruction of hypoplastic left heart syndrome and variants. J Thorac Cardiovasc Surg. 2008;136:94–9. - PubMed
-
- Furck AK, Uebing A, Hansen JH, Scheewe J, Jung O, Fischer G, et al. Outcome of the Norwood operation in patients with hypoplastic left heart syndrome: a 12-year single-center survey. J Thorac Cardiovasc Surg. 2010;139:359–65. - PubMed
-
- Jonas RA, Hansen DD, Cook N, Wessel D. Anatomic subtype and survival after reconstructive operation for hypoplastic left heart syndrome. J Thorac Cardiovasc Surg. 1994;107:1121–8. - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
- HL068288/HL/NHLBI NIH HHS/United States
- HL068285/HL/NHLBI NIH HHS/United States
- U01 HL068269/HL/NHLBI NIH HHS/United States
- U01 HL068279/HL/NHLBI NIH HHS/United States
- U01 HL068290/HL/NHLBI NIH HHS/United States
- U10 HL068270/HL/NHLBI NIH HHS/United States
- U01 HL068281/HL/NHLBI NIH HHS/United States
- U01 HL085057/HL/NHLBI NIH HHS/United States
- HL068290/HL/NHLBI NIH HHS/United States
- HL068279/HL/NHLBI NIH HHS/United States
- U01 HL068288/HL/NHLBI NIH HHS/United States
- U01 HL068270/HL/NHLBI NIH HHS/United States
- HL085057/HL/NHLBI NIH HHS/United States
- U01 HL068292/HL/NHLBI NIH HHS/United States
- HL068269/HL/NHLBI NIH HHS/United States
- HL068270/HL/NHLBI NIH HHS/United States
- U01 HL068285/HL/NHLBI NIH HHS/United States
- HL068292/HL/NHLBI NIH HHS/United States

