Long-term mechanical circulatory support (destination therapy): on track to compete with heart transplantation?
- PMID: 22795459
- PMCID: PMC3443856
- DOI: 10.1016/j.jtcvs.2012.05.044
Long-term mechanical circulatory support (destination therapy): on track to compete with heart transplantation?
Abstract
Objectives: Average 2-year survival after cardiac transplantation is approximately 80%. The evolution and subsequent approval of larger pulsatile and, more recently, continuous flow mechanical circulatory support (MCS) technology for destination therapy (DT) offers the potential for triage of some patients awaiting cardiac transplantation to DT.
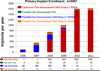
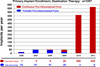
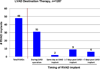
Methods: The National Heart, Lung, and Blood Institute Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) is a national multi-institutional study of long-term MCS. Between June 2006 and December 2011, 127 pulsatile and 1160 continuous flow pumps (24% of total primary left ventricular assist devices [LVADs]) carried an initial strategy of DT therapy.
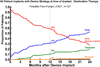
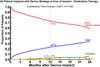
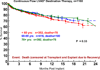
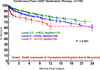
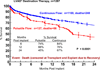
Results: By multivariable analysis, risk factors (P < .05) for mortality after DT included older age, larger body mass index, history of cancer, history of cardiac surgery, INTERMACS level I (cardiogenic shock), dialysis, increased blood urea nitrogen, use of a pulsatile flow device, and use of a right ventricular assist device (RVAD). Among patients with a continuous flow LVAD who were not in cardiogenic shock, a particularly favorable survival was associated with no cancer, patients not in cardiogenic shock, and blood urea nitrogen less than 50 mg/dL, resulting in 1- and 2-year survivals of 88% and 80%.
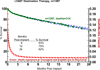
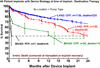
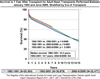
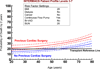
Conclusions: (1) Evolution from pulsatile to continuous flow technology has dramatically improved 1- and 2-year survivals; (2) DT is not appropriate for patients with rapid hemodynamic deterioration or severe right ventricular failure; (3) important subsets of patients with continuous flow DT now enjoy survival that is competitive with heart transplantation out to about 2 years.
Copyright © 2012 The American Association for Thoracic Surgery. Published by Mosby, Inc. All rights reserved.
Figures







References
-
- Miller MA, Ulisney K, Baldwin JT, et al. INTERMACS (Interagency Registry for Mechanically Assisted Circulatory Support): A New Paradigm for Translating Registry Data Into Clinical Practice. Journal of American College of Cardiology. 2010;56(9):738–740. - PubMed
-
- Kirklin JK, Naftel DC, Stevenson LW, et al. INTERMACS Database for Durable Devices for Circulatory Support: First Annual Report. J Heart Lung Transplant. 2008;27:1065–1072. - PubMed
-
- Warner-Stevenson L, Kirklin JK, Pagani FD, et al. INTERMACS Profiles of Advanced Heart Failure: First Definition. J Heart Lung Transp. 2009;28:535–541. - PubMed
-
- McGiffin DC, Naftel DC, Kirklin JK, et al. Predicting Outcome After Listing for Heart Transplantation in Children: Comparison of Kaplan-Meier and Parametric Competing Risk Analysis. J Heart Lung Transplant. 1997;16:713–722. - PubMed
-
- Stehlik J, Edwards lB, Kucheryavaya AY, et al. The Registry of the International Society for Heart and Lung Transplantation: Twenty-eighth Adult Heart Transplant Report-2011. J Heart Lung Transplant. 2011;30(10):1078–1094. - PubMed

