Anti-CD20 multivalent HPMA copolymer-Fab' conjugates for the direct induction of apoptosis
- PMID: 22795544
- PMCID: PMC3572842
- DOI: 10.1016/j.biomaterials.2012.06.024
Anti-CD20 multivalent HPMA copolymer-Fab' conjugates for the direct induction of apoptosis
Abstract
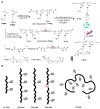
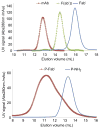
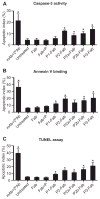
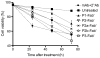
A hybrid biomimetic system comprising high-molecular-weight, linear copolymer of N-(2-hydroxypropyl)methacrylamide (HPMA) grafted with multiple Fab' fragments of anti-CD20 monoclonal antibody (mAb) was synthesized by reversible addition-fragmentation chain transfer (RAFT) polymerization followed by attachment of Fab' fragments via thioether bonds. Exposure of human non-Hodgkin's lymphoma (NHL) Raji B cells to the multivalent conjugates resulted in crosslinking of CD20 receptors and commencement of apoptosis. Five conjugates with varying molecular weight and valence (amount of Fab' per polymer chain) were synthesized. One of the copolymers contained enzyme degradable peptide sequences (GFLG) in the backbone. The multivalency led to higher avidity and apoptosis induction compared to unconjugated whole mAb. Time-dependent studies showed that the cytotoxicity of conjugates exhibited a slower onset at shorter exposure times than mAb hyper-crosslinked with a secondary Ab; however, at longer time intervals the HPMA copolymer conjugates achieved significantly higher biological efficacies. In addition, study of the relationship between the structure of conjugates and Raji B cell apoptosis revealed that both valency and polymer molecular weight influenced biological activities, while insertion of peptide sequences into the backbone was not a factor in vitro.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
Biological activity of anti-CD20 multivalent HPMA copolymer-Fab' conjugates.Biomacromolecules. 2012 Mar 12;13(3):727-35. doi: 10.1021/bm201656k. Epub 2012 Feb 21. Biomacromolecules. 2012. PMID: 22288884 Free PMC article.
-
Drug-free macromolecular therapeutics: Impact of structure on induction of apoptosis in Raji B cells.J Control Release. 2017 Oct 10;263:139-150. doi: 10.1016/j.jconrel.2016.12.025. Epub 2016 Dec 24. J Control Release. 2017. PMID: 28024916 Free PMC article.
-
Synthesis and evaluation of multivalent branched HPMA copolymer-Fab' conjugates targeted to the B-cell antigen CD20.Bioconjug Chem. 2009 Jan;20(1):129-37. doi: 10.1021/bc800351m. Bioconjug Chem. 2009. PMID: 19154157 Free PMC article.
-
What signals are generated by anti-CD20 antibody therapy?Curr Hematol Malig Rep. 2006 Dec;1(4):205-13. doi: 10.1007/s11899-006-0001-z. Curr Hematol Malig Rep. 2006. PMID: 20425315 Review.
-
Design of smart HPMA copolymer-based nanomedicines.J Control Release. 2016 Oct 28;240:9-23. doi: 10.1016/j.jconrel.2015.10.003. Epub 2015 Oct 3. J Control Release. 2016. PMID: 26437260 Free PMC article. Review.
Cited by
-
Multi-targeted immunotherapeutics to treat B cell malignancies.J Control Release. 2023 Jun;358:232-258. doi: 10.1016/j.jconrel.2023.04.048. Epub 2023 May 5. J Control Release. 2023. PMID: 37121515 Free PMC article. Review.
-
Hydrophilic biomaterials: From crosslinked and self-assembled hydrogels to polymer-drug conjugates and drug-free macromolecular therapeutics.J Control Release. 2024 Sep;373:1-22. doi: 10.1016/j.jconrel.2024.05.012. Epub 2024 May 17. J Control Release. 2024. PMID: 38734315
-
Polymer-drug conjugate therapeutics: advances, insights and prospects.Nat Rev Drug Discov. 2019 Apr;18(4):273-294. doi: 10.1038/s41573-018-0005-0. Nat Rev Drug Discov. 2019. PMID: 30542076 Free PMC article. Review.
-
Tracking and quantifying polymer therapeutic distribution on a cellular level using 3D dSTORM.J Control Release. 2016 Jun 10;231:50-9. doi: 10.1016/j.jconrel.2016.02.005. Epub 2016 Feb 16. J Control Release. 2016. PMID: 26855050 Free PMC article.
-
Neurosurgery at the crossroads of immunology and nanotechnology. New reality in the COVID-19 pandemic.Adv Drug Deliv Rev. 2022 Feb;181:114033. doi: 10.1016/j.addr.2021.114033. Epub 2021 Nov 20. Adv Drug Deliv Rev. 2022. PMID: 34808227 Free PMC article. Review.
References
-
- Couvreur P, Vauthier C. Nanotechnology: intelligent design to treat complex disease. Pharm Res. 2006;23(7):1417–50. - PubMed
-
- Kopeček J. Smart and genetically engineered biomaterials and drug delivery systems. Eur J Pharm Sci. 2003;20(1):1–16. - PubMed
-
- Ulbrich K, Šubr V. Structural and chemical aspects of HPMA copolymers as drug carriers. Adv Drug Deliv Rev. 2010;62(2):150–66. - PubMed
-
- Vicent MJ, Ringsdorf H, Duncan R. Polymer therapeutics: clinical applications and challenges for development. Adv Drug Deliv Rev. 2009;61(13):1117–20. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

