Paracrine signaling through plasma membrane hemichannels
- PMID: 22796188
- PMCID: PMC3666170
- DOI: 10.1016/j.bbamem.2012.07.002
Paracrine signaling through plasma membrane hemichannels
Abstract
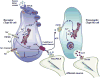
Plasma membrane hemichannels composed of connexin (Cx) proteins are essential components of gap junction channels but accumulating evidence suggests functions of hemichannels beyond the communication provided by junctional channels. Hemichannels not incorporated into gap junctions, called unapposed hemichannels, can open in response to a variety of signals, electrical and chemical, thereby forming a conduit between the cell's interior and the extracellular milieu. Open hemichannels allow the bidirectional passage of ions and small metabolic or signaling molecules of below 1-2kDa molecular weight. In addition to connexins, hemichannels can also be formed by pannexin (Panx) proteins and current evidence suggests that Cx26, Cx32, Cx36, Cx43 and Panx1, form hemichannels that allow the diffusive release of paracrine messengers. In particular, the case is strong for ATP but substantial evidence is also available for other messengers like glutamate and prostaglandins or metabolic substances like NAD(+) or glutathione. While this field is clearly in expansion, evidence is still lacking at essential points of the paracrine signaling cascade that includes not only messenger release, but also downstream receptor signaling and consequent functional effects. The data available at this moment largely derives from in vitro experiments and still suffers from the difficulty of separating the functions of connexin-based hemichannels from gap junctions and from pannexin hemichannels. However, messengers like ATP or glutamate have universal roles in the body and further defining the contribution of hemichannels as a possible release pathway is expected to open novel avenues for better understanding their contribution to a variety of physiological and pathological processes. This article is part of a Special Issue entitled: The Communicating junctions, roles and dysfunctions.
Copyright © 2012 Elsevier B.V. All rights reserved.
Figures
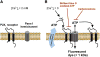

References
-
- Goodenough DA, Goliger JA, Paul DL. Connexins, connexons, and intercellular communication. Annu Rev Biochem. 1996;65:475–502. - PubMed
-
- Willecke K, Eiberger J, Degen J, Eckardt D, Romualdi A, Guldenagel M, Deutsch U, Sohl G. Structural and functional diversity of connexin genes in the mouse and human genome. Biol Chem. 2002;383:725–737. - PubMed
-
- Saez JC, Berthoud VM, Branes MC, Martinez AD, Beyer EC. Plasma membrane channels formed by connexins: their regulation and functions. Physiol Rev. 2003;83:1359–1400. - PubMed
-
- Bruzzone R, White TW, Paul DL. Connections with connexins: the molecular basis of direct intercellular signaling. Eur J Biochem. 1996;238:1–27. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

