Proline dehydrogenase is essential for proline protection against hydrogen peroxide-induced cell death
- PMID: 22796327
- PMCID: PMC3432146
- DOI: 10.1016/j.freeradbiomed.2012.07.002
Proline dehydrogenase is essential for proline protection against hydrogen peroxide-induced cell death
Abstract
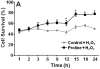
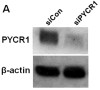
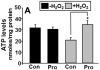
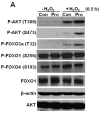
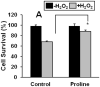
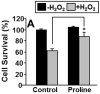
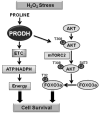
Proline metabolism has an underlying role in apoptotic signaling that influences tumorigenesis. Proline is oxidized to glutamate in the mitochondria, with the rate-limiting step catalyzed by proline dehydrogenase (PRODH). PRODH expression is inducible by p53, leading to increased proline oxidation, reactive oxygen species formation, and induction of apoptosis. Paradoxical to its role in apoptosis, proline also protects cells against oxidative stress. Here we explore the mechanism of proline protection against hydrogen peroxide stress in melanoma WM35 cells. Treatment of WM35 cells with proline significantly increased cell viability, diminished oxidative damage of cellular lipids and proteins, and maintained ATP and NADPH levels after exposure to hydrogen peroxide. Inhibition or siRNA-mediated knockdown of PRODH abolished proline protection against oxidative stress, whereas knockdown of Δ(1)-pyrroline-5-carboxylate reductase, a key enzyme in proline biosynthesis, had no impact on proline protection. Potential linkages between proline metabolism and signaling pathways were explored. The combined inhibition of the mammalian target of rapamycin complex 1 (mTORC1) and mTORC2 eliminated proline protection. A significant increase in Akt activation was observed in proline-treated cells after hydrogen peroxide stress along with a corresponding increase in the phosphorylation of the forkhead transcription factor class O3a (FoxO3a). The role of PRODH in proline-mediated protection was validated in the prostate carcinoma cell line PC3. Knockdown of PRODH in PC3 cells attenuated phosphorylated levels of Akt and FoxO3a and decreased cell survival during hydrogen peroxide stress. The results provide evidence that PRODH is essential in proline protection against hydrogen peroxide-mediated cell death and that proline/PRODH helps activate Akt in cancer cells.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures




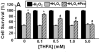
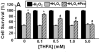
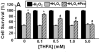
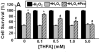




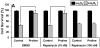
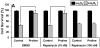













References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

