Multimeric structures of HLA-G isoforms function through differential binding to LILRB receptors
- PMID: 22802125
- PMCID: PMC3884069
- DOI: 10.1007/s00018-012-1069-3
Multimeric structures of HLA-G isoforms function through differential binding to LILRB receptors
Abstract
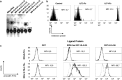
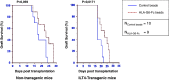
The non-classical Human leukocyte antigen G (HLA-G) differs from classical HLA class I molecules by its low genetic diversity, a tissue-restricted expression, the existence of seven isoforms, and immuno-inhibitory functions. Most of the known functions of HLA-G concern the membrane-bound HLA-G1 and soluble HLA-G5 isoforms, which present the typical structure of classical HLA class I molecule: a heavy chain of three globular domains α1-α2-α3 non-covalently bound to β-2-microglobulin (B2M) and a peptide. Very little is known of the structural features and functions of other HLA-G isoforms or structural conformations other than B2M-associated HLA-G1 and HLA-G5. In the present work, we studied the capability of all isoforms to form homomultimers, and investigated whether they could bind to, and function through, the known HLA-G receptors LILRB1 and LILRB2. We report that all HLA-G isoforms may form homodimers, demonstrating for the first time the existence of HLA-G4 dimers. We also report that the HLA-G α1-α3 structure, which constitutes the extracellular part of HLA-G2 and HLA-G6, binds the LILRB2 receptor but not LILRB1. This is the first report of a receptor for a truncated HLA-G isoform. Following up on this finding, we show that the α1-α3-Fc structure coated on agarose beads is tolerogenic and capable of prolonging the survival of skin allografts in B6-mice and in a LILRB2-transgenic mouse model. This study is the first proof of concept that truncated HLA-G isoforms could be used as therapeutic agents.
Figures


Similar articles
-
Enhanced effect of the immunosuppressive soluble HLA-G2 homodimer by site-specific PEGylation.Sci Rep. 2025 Jan 20;15(1):2509. doi: 10.1038/s41598-024-85072-x. Sci Rep. 2025. PMID: 39833185 Free PMC article.
-
Structural basis for recognition of the nonclassical MHC molecule HLA-G by the leukocyte Ig-like receptor B2 (LILRB2/LIR2/ILT4/CD85d).Proc Natl Acad Sci U S A. 2006 Oct 31;103(44):16412-7. doi: 10.1073/pnas.0605228103. Epub 2006 Oct 20. Proc Natl Acad Sci U S A. 2006. PMID: 17056715 Free PMC article.
-
Structural and Functional Basis for LILRB Immune Checkpoint Receptor Recognition of HLA-G Isoforms.J Immunol. 2019 Dec 15;203(12):3386-3394. doi: 10.4049/jimmunol.1900562. Epub 2019 Nov 6. J Immunol. 2019. PMID: 31694909
-
Soluble monomers, dimers and HLA-G-expressing extracellular vesicles: the three dimensions of structural complexity to use HLA-G as a clinical biomarker.HLA. 2016 Sep;88(3):77-86. doi: 10.1111/tan.12844. Epub 2016 Jul 21. HLA. 2016. PMID: 27440734 Review.
-
HLA-G modulates immune responses by diverse receptor interactions.Semin Cancer Biol. 2003 Oct;13(5):317-23. doi: 10.1016/s1044-579x(03)00022-1. Semin Cancer Biol. 2003. PMID: 14708711 Review.
Cited by
-
First immunotherapeutic CAR-T cells against the immune checkpoint protein HLA-G.J Immunother Cancer. 2021 Mar;9(3):e001998. doi: 10.1136/jitc-2020-001998. J Immunother Cancer. 2021. PMID: 33737343 Free PMC article.
-
Therapeutic Effects of HLA-G5 Overexpressing hAMSCs on aGVHD After Allo-HSCT: Involving in the Gut Microbiota at the Intestinal Barrier.J Inflamm Res. 2023 Aug 24;16:3669-3685. doi: 10.2147/JIR.S420747. eCollection 2023. J Inflamm Res. 2023. PMID: 37645691 Free PMC article.
-
Soluble HLA-G expression levels and HLA-G/irinotecan association in metastatic colorectal cancer treated with irinotecan-based strategy.Sci Rep. 2020 May 29;10(1):8773. doi: 10.1038/s41598-020-65424-z. Sci Rep. 2020. PMID: 32471996 Free PMC article.
-
The Molecular and Functional Characteristics of HLA-G and the Interaction with Its Receptors: Where to Intervene for Cancer Immunotherapy?Int J Mol Sci. 2020 Nov 17;21(22):8678. doi: 10.3390/ijms21228678. Int J Mol Sci. 2020. PMID: 33213057 Free PMC article. Review.
-
HLA-G Neo-Expression on Tumors.Front Immunol. 2020 Aug 14;11:1685. doi: 10.3389/fimmu.2020.01685. eCollection 2020. Front Immunol. 2020. PMID: 32922387 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

