Time course of inflammation, oxidative stress and tissue damage induced by hyperoxia in mouse lungs
- PMID: 22804763
- PMCID: PMC3444983
- DOI: 10.1111/j.1365-2613.2012.00823.x
Time course of inflammation, oxidative stress and tissue damage induced by hyperoxia in mouse lungs
Abstract
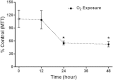
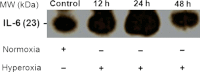
In this study our aim was to investigate the time courses of inflammation, oxidative stress and tissue damage after hyperoxia in the mouse lung. Groups of BALB/c mice were exposed to 100% oxygen in a chamber for 12, 24 or 48 h. The controls were subjected to normoxia. The results showed that IL-6 increased progressively after 12 (P < 0.001) and 24 h (P < 0.001) of hyperoxia with a reduction at 48 h (P < 0.01), whereas TNF-α increased after 24 (P < 0.001) and 48 h (P < 0.001). The number of macrophages increased after 24 h (P < 0.001), whereas the number of neutrophils increased after 24 h (P < 0.01) and 48 h (P < 0.001). Superoxide dismutase activity decreased in all groups exposed to hyperoxia (P < 0.01). Catalase activity increased only at 48 h (P < 0.001). The reduced glutathione/oxidized glutathione ratio decreased after 12 h (P < 0.01) and 24 h (P < 0.05). Histological evidence of lung injury was observed at 24 and 48 h. This study shows that hyperoxia initially causes an inflammatory response at 12 h, resulting in inflammation associated with the oxidative response at 24 h and culminating in histological damage at 48 h. Knowledge of the time course of inflammation and oxidative stress prior to histological evidence of acute lung injury can improve the safety of oxygen therapy in patients.
© 2012 The Authors. International Journal of Experimental Pathology © 2012 International Journal of Experimental Pathology.
Figures




References
-
- Aebi H. Catalase in vitro. Methods Enzymol. 1984;105:121–126. - PubMed
-
- Alejandre-Alcazar MA, Kwapiszewska G, Reiss I, et al. Hyperoxia modulates TGF-beta/BMP signaling in a mouse model of bronchopulmonary dysplasia. Am. J. Physiol. 2007;292:L537–L549. - PubMed
-
- Bannister JV, Calabrese L. Assays for superoxide dismutase. Methods Biochem. Anal. 1987;32:279–312. - PubMed
-
- Bhandari V. Molecular mechanisms of hyperoxia-induced acute lung injury. Front. Biosci. 2008;13:6653–6661. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

