Perilipin 2 improves insulin sensitivity in skeletal muscle despite elevated intramuscular lipid levels
- PMID: 22807032
- PMCID: PMC3478528
- DOI: 10.2337/db11-1402
Perilipin 2 improves insulin sensitivity in skeletal muscle despite elevated intramuscular lipid levels
Abstract
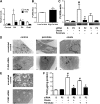
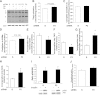
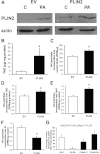
Type 2 diabetes is characterized by excessive lipid storage in skeletal muscle. Excessive intramyocellular lipid (IMCL) storage exceeds intracellular needs and induces lipotoxic events, ultimately contributing to the development of insulin resistance. Lipid droplet (LD)-coating proteins may control proper lipid storage in skeletal muscle. Perilipin 2 (PLIN2/adipose differentiation-related protein [ADRP]) is one of the most abundantly expressed LD-coating proteins in skeletal muscle. Here we examined the role of PLIN2 in myocellular lipid handling and insulin sensitivity by investigating the effects of in vitro PLIN2 knockdown and in vitro and in vivo overexpression. PLIN2 knockdown decreased LD formation and triacylglycerol (TAG) storage, marginally increased fatty-acid (FA) oxidation, and increased incorporation of palmitate into diacylglycerols and phospholipids. PLIN2 overexpression in vitro increased intramyocellular TAG storage paralleled with improved insulin sensitivity. In vivo muscle-specific PLIN2 overexpression resulted in increased LD accumulation and blunted the high-fat diet-induced increase in protein content of the subunits of the oxidative phosphorylation (OXPHOS) chain. Diacylglycerol levels were unchanged, whereas ceramide levels were increased. Despite the increased IMCL accumulation, PLIN2 overexpression improved skeletal muscle insulin sensitivity. We conclude that PLIN2 is essential for lipid storage in skeletal muscle by enhancing the partitioning of excess FAs toward TAG storage in LDs, thereby blunting lipotoxicity-associated insulin resistance.
Figures


References
-
- Goodpaster BH, Wolf D. Skeletal muscle lipid accumulation in obesity, insulin resistance, and type 2 diabetes. Pediatr Diabetes 2004;5:219–226 - PubMed
-
- Goodpaster BH, He J, Watkins S, Kelley DE. Skeletal muscle lipid content and insulin resistance: evidence for a paradox in endurance-trained athletes. J Clin Endocrinol Metab 2001;86:5755–5761 - PubMed
-
- van Loon LJC, Goodpaster BH. Increased intramuscular lipid storage in the insulin-resistant and endurance-trained state. Pflugers Arch 2006;451:606–616 - PubMed
-
- Bruce CR, Anderson MJ, Carey AL, et al. Muscle oxidative capacity is a better predictor of insulin sensitivity than lipid status. J Clin Endocrinol Metab 2003;88:5444–5451 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

