Structure and functional studies of N-terminal Cx43 mutants linked to oculodentodigital dysplasia
- PMID: 22809623
- PMCID: PMC3431933
- DOI: 10.1091/mbc.E12-02-0128
Structure and functional studies of N-terminal Cx43 mutants linked to oculodentodigital dysplasia
Abstract
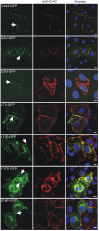
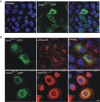
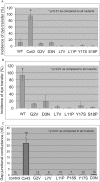
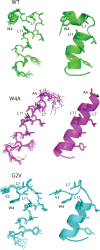
Mutations in the gene encoding connexin-43 (Cx43) cause the human development disorder known as oculodentodigital dysplasia (ODDD). In this study, ODDD-linked Cx43 N-terminal mutants formed nonfunctional gap junction-like plaques and exhibited dominant-negative effects on the coupling conductance of coexpressed endogenous Cx43 in reference cell models. Nuclear magnetic resonance (NMR) protein structure determination of an N-terminal 23-amino acid polypeptide of wild-type Cx43 revealed that it folded in to a kinked α-helical structure. This finding predicted that W4 might be critically important in intramolecular and intermolecular interactions. Thus we engineered and characterized a W4A mutant and found that this mutant formed a regular, nonkinked α-helix but did not form functional gap junctions. Furthermore, a G2V variant peptide of Cx43 showed a kinked helix that now included V2 interactions with W4, resulting in the G2V mutant forming nonfunctional gap junctions. Also predicted from the NMR structures, a G2S mutant was found to relieve these interactions and allowed the protein to form functional gap junctions. Collectively, these studies suggest that the nature of the mutation conveys loss of Cx43 function by distinctly different mechanisms that are rooted in the structure of the N-terminal region.
Figures
Similar articles
-
Autosomal recessive GJA1 (Cx43) gene mutations cause oculodentodigital dysplasia by distinct mechanisms.J Cell Sci. 2013 Jul 1;126(Pt 13):2857-66. doi: 10.1242/jcs.123315. Epub 2013 Apr 19. J Cell Sci. 2013. PMID: 23606748
-
Syndromic and non-syndromic disease-linked Cx43 mutations.FEBS Lett. 2014 Apr 17;588(8):1339-48. doi: 10.1016/j.febslet.2013.12.022. Epub 2014 Jan 14. FEBS Lett. 2014. PMID: 24434540 Review.
-
Manipulating Cx43 expression triggers gene reprogramming events in dermal fibroblasts from oculodentodigital dysplasia patients.Biochem J. 2015 Nov 15;472(1):55-69. doi: 10.1042/BJ20150652. Epub 2015 Sep 8. Biochem J. 2015. PMID: 26349540
-
Specific functional pathologies of Cx43 mutations associated with oculodentodigital dysplasia.Mol Biol Cell. 2016 Jul 15;27(14):2172-85. doi: 10.1091/mbc.E16-01-0062. Epub 2016 May 25. Mol Biol Cell. 2016. PMID: 27226478 Free PMC article.
-
Oculodentodigital Dysplasia with a Novel Mutation in GJA1 Diagnosed by Targeted Gene Panel Sequencing: A Case Report and Literature Review.Ann Clin Lab Sci. 2018 Nov;48(6):776-781. Ann Clin Lab Sci. 2018. PMID: 30610049 Review.
Cited by
-
Cryo-EM structures of undocked innexin-6 hemichannels in phospholipids.Sci Adv. 2020 Feb 12;6(7):eaax3157. doi: 10.1126/sciadv.aax3157. eCollection 2020 Feb. Sci Adv. 2020. PMID: 32095518 Free PMC article.
-
Towards a Better Understanding of Genotype-Phenotype Correlations and Therapeutic Targets for Cardiocutaneous Genes: The Importance of Functional Studies above Prediction.Int J Mol Sci. 2022 Sep 15;23(18):10765. doi: 10.3390/ijms231810765. Int J Mol Sci. 2022. PMID: 36142674 Free PMC article. Review.
-
Structure of the connexin-43 gap junction channel in a putative closed state.Elife. 2023 Aug 3;12:RP87616. doi: 10.7554/eLife.87616. Elife. 2023. PMID: 37535063 Free PMC article.
-
Determinants of Cx43 Channel Gating and Permeation: The Amino Terminus.Biophys J. 2016 Jan 5;110(1):127-40. doi: 10.1016/j.bpj.2015.10.054. Biophys J. 2016. PMID: 26745416 Free PMC article.
-
Connexins in Cardiovascular and Neurovascular Health and Disease: Pharmacological Implications.Pharmacol Rev. 2017 Oct;69(4):396-478. doi: 10.1124/pr.115.012062. Pharmacol Rev. 2017. PMID: 28931622 Free PMC article. Review.
References
-
- Arita K, Akiyama M, Aizawa T, Umetsu Y, Segawa I, Goto M, Sawamura D, Demura M, Kawano K, Shimizu H. A novel N14Y mutation in connexin26 in keratitis-ichthyosis-deafness syndrome: analyses of altered gap junctional communication and molecular structure of N terminus of mutated connexin26. Am J Pathol. 2006;169:416–423. - PMC - PubMed
-
- Bax A, Davis DG. MLEV-17 based two-dimensional homonuclear magetization transfer spectroscopy. J Magn Reson. 1985;65:355–360.
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

