A pilot study evaluating the safety and toxicity of epirubicin, cisplatin, and UFT (ECU regimen) in advanced gastric carcinoma
- PMID: 22811823
- PMCID: PMC3397594
- DOI: 10.3978/j.issn.2078-6891.2010.030
A pilot study evaluating the safety and toxicity of epirubicin, cisplatin, and UFT (ECU regimen) in advanced gastric carcinoma
Abstract
Background: Best response rates have been achieved with three-drug regimens containing 5-FU in the treatment of advanced gastric cancer (AGC) and oral fluoropyrimidines are the best alternatives as substitutes for infusional 5-FU. This study aimed to evaluate the safety and toxicity of epirubicin, cisplatin, and UFT (ECU regimen) regimens in AGC outpatients.
Materials and methods: Forty-one patients with AGC received epirubicin, cisplatin, and oral UFT plus leucovorin. Epirubicin 50 mg/m(2) and cisplatin 60mg/m(2) were administered on Day 1. Three hundreds (300) mg/m(2)/day UFT was administered with leucovorin at a fixed oral dose of 90 mg/day for 21 days, followed by a 7-day rest period. Cycles were repeated every 4 weeks. Performance status was either as 0 and 1.
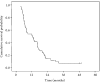
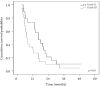
Results: Among the 41 patients enrolled, complete and partial response was achieved in 7.3% and 36.6% of patients, respectively, with an overall response rate of 43.9%. Stable disease was observed in 34.1% of patients and 22% showed disease progression. Median time to progression was 5.2 months and median survival was 12.3 months. A median of 4 cycles (range: 1-6) of chemotherapy were administered. The main grade III-IV toxicities were nausea/vomiting (19.4%) and neutropenia (12.1%). Grade IV toxicities were gastric perforation and renal failure.
Conclusion: ECU appears to be an effective regimen in the treatment of AGC, with acceptable tolerability and manageable toxicity. In three-drug regimens, substitution of infusional 5-FU by UFT offers the possibility of increased AGC outpatient compliance.
Keywords: UFT; advanced gastric carcinoma; cisplatin; epirubicin.
Conflict of interest statement
No potential conflict of interest.
Figures
References
-
- Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108. - PubMed
-
- Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics, 2007. CA Cancer J Clin. 2007;57:43–66. - PubMed
-
- Sengelen M, Kutluk T, Fırat D. Cancer Statistics in Turkey and in the World 1996-2003. 1st edition Turkish Association for Cancer Research and Control. 2007:p46.
-
- Demir G, Zengin N, Yıldız O. Clinical profile of gastric cancer in Turkey: Factors effecting disease free survival and relapse (Turkish Oncology Group Study) Annals of Oncology. 2006;17 Abtract :1139 and updated, unpublished data with personal communication.
-
- Maruyama K, Kaminishi M, Hayashi K, Isobe Y, Honda I, Katai H, et al. Gastric cancer treated in 1991 in Japan: data analysis of nationwide registry. Gastric Cancer. 2006;9:51–66. - PubMed
LinkOut - more resources
Full Text Sources