Abnormal vascularization in mouse retina with dysregulated retinal cholesterol homeostasis
- PMID: 22820291
- PMCID: PMC3408752
- DOI: 10.1172/JCI63816
Abnormal vascularization in mouse retina with dysregulated retinal cholesterol homeostasis
Abstract
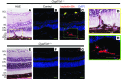
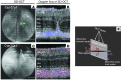
Several lines of evidence suggest a link between age-related macular degeneration and retinal cholesterol maintenance. Cytochrome P450 27A1 (CYP27A1) is a ubiquitously expressed mitochondrial sterol 27-hydroxylase that plays an important role in the metabolism of cholesterol and cholesterol-related compounds. We conducted a comprehensive ophthalmic evaluation of mice lacking CYP27A1. We found that the loss of CYP27A1 led to dysregulation of retinal cholesterol homeostasis, including unexpected upregulation of retinal cholesterol biosynthesis. Cyp27a1-/- mice developed retinal lesions characterized by cholesterol deposition beneath the retinal pigment epithelium. Further, Cyp27a1-null mice showed pathological neovascularization, which likely arose from both the retina and the choroid, that led to the formation of retinal-choroidal anastomosis. Blood flow alterations and blood vessel leakage were noted in the areas of pathology. The Cyp27a1-/- retina was hypoxic and had activated Müller cells. We suggest a mechanism whereby abolished sterol 27-hydroxylase activity leads to vascular changes and identify Cyp27a1-/- mice as a model for one of the variants of type 3 retinal neovascularization occurring in some patients with age-related macular degeneration.
Figures








References
-
- Gass JD. Atlas of Macular Diseases. St. Louis, Missouri, USA: CV Mosby; 1997.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

