Persistent astroglial swelling accompanies rapid reversible dendritic injury during stroke-induced spreading depolarizations
- PMID: 22821441
- PMCID: PMC3435464
- DOI: 10.1002/glia.22390
Persistent astroglial swelling accompanies rapid reversible dendritic injury during stroke-induced spreading depolarizations
Abstract
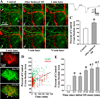
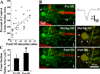
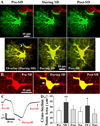
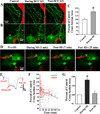
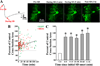
Spreading depolarizations are a key event in the pathophysiology of stroke, resulting in rapid dendritic beading, which represents acute damage to synaptic circuitry. The impact of spreading depolarizations on the real-time injury of astrocytes during ischemia is less clear. We used simultaneous in vivo 2-photon imaging and electrophysiological recordings in adult mouse somatosensory cortex to examine spreading depolarization-induced astroglial structural changes concurrently with signs of neuronal injury in the early periods of focal and global ischemia. Astrocytes in the metabolically compromised ischemic penumbra-like area showed a long lasting swelling response to spontaneous spreading depolarizations despite rapid dendritic recovery in a photothrombotic occlusion model of focal stroke. Astroglial swelling was often facilitated by recurrent depolarizations and the magnitude of swelling strongly correlated with the total duration of depolarization. In contrast, spreading depolarization-induced astroglial swelling was transient in normoxic healthy tissue. In a model of transient global ischemia, the occurrence of a single spreading depolarization elicited by a bilateral common carotid artery occlusion coincided with astroglial swelling alongside dendritic beading. With immediate reperfusion, dendritic beading subsides. Astroglial swelling was either transient during short ischemic periods distinguished by a short-lasting spreading depolarization, or persistent during severe ischemia characterized by a long-lasting depolarization with the ultraslow negative voltage component. We propose that persistent astroglial swelling is initiated and exacerbated during spreading depolarization in brain tissue with moderate to severe energy deficits, disrupting astroglial maintenance of normal homeostatic function thus contributing to the negative outcome of ischemic stroke as astrocytes fail to provide neuronal support.
Copyright © 2012 Wiley Periodicals, Inc.
Figures
References
-
- Aitken PG, Tombaugh GC, Turner DA, Somjen GG. Similar propagation of SD and hypoxic SD-like depolarization in rat hippocampus recorded optically and electrically. J Neurophysiol. 1998;80:1514–1521. - PubMed
-
- Amiry-Moghaddam M, Ottersen OP. The molecular basis of water transport in the brain. Nat Rev Neurosci. 2003;4:991–1001. - PubMed
-
- Andrew RD, Labron MW, Boehnke SE, Carnduff L, Kirov SA. Physiological evidence that pyramidal neurons lack functional water channels. Cereb Cortex. 2007;17:787–802. - PubMed
-
- Araque A, Parpura V, Sanzgiri RP, Haydon PG. Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci. 1999;22:208–215. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

