Enhanced antigen retrieval of amyloid β immunohistochemistry: re-evaluation of amyloid β pathology in Alzheimer disease and its mouse model
- PMID: 22821668
- PMCID: PMC3524564
- DOI: 10.1369/0022155412456379
Enhanced antigen retrieval of amyloid β immunohistochemistry: re-evaluation of amyloid β pathology in Alzheimer disease and its mouse model
Abstract
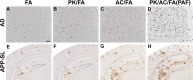
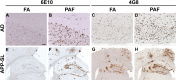
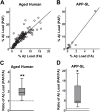
Senile plaques, extracellular deposits of amyloid β peptide (Aβ), are one of the pathological hallmarks of Alzheimer disease (AD). As the standard immunohistochemical detection method for Aβ deposits, anti-Aβ immunohistochemistry combined with antigen retrieval (AR) by formic acid (FA) has been generally used. Here, we present a more efficient AR for Aβ antigen. On brain sections of AD and its mouse model, a double combination of either autoclave heating in EDTA buffer or digestion with proteinase K plus FA treatment reinforced Aβ immunoreactivity. A further triple combination of digestion with proteinase K (P), autoclave heating in EDTA buffer (A), and FA treatment (F), when employed in this order, gave a more enhanced immunoreactivity. Our PAF method prominently visualized various forms of Aβ deposits in AD that have not been clearly detected previously and revealed numerous minute-sized plaques both in AD and the mouse model. Quantification of Aβ loads showed that the AR effect by the PAF method was 1.86-fold (in the aged human brain) and 4.64-fold (in the mouse brain) higher than that by the FA method. Thus, the PAF method could have the potential to be the most sensitive tool so far to study Aβ pathology in AD and its mouse model.
Conflict of interest statement
Figures

Similar articles
-
Pyroglutamate-3 amyloid-β deposition in the brains of humans, non-human primates, canines, and Alzheimer disease-like transgenic mouse models.Am J Pathol. 2013 Aug;183(2):369-81. doi: 10.1016/j.ajpath.2013.05.005. Epub 2013 Jun 7. Am J Pathol. 2013. PMID: 23747948 Free PMC article.
-
High sensitivity analysis of amyloid-beta peptide composition in amyloid deposits from human and PS2APP mouse brain.Neuroscience. 2006 Dec 1;143(2):461-75. doi: 10.1016/j.neuroscience.2006.08.027. Epub 2006 Sep 27. Neuroscience. 2006. PMID: 17008022
-
N-truncated Aβ4-x peptides in sporadic Alzheimer's disease cases and transgenic Alzheimer mouse models.Alzheimers Res Ther. 2017 Oct 4;9(1):80. doi: 10.1186/s13195-017-0309-z. Alzheimers Res Ther. 2017. PMID: 28978359 Free PMC article.
-
Alzheimer's disease.Subcell Biochem. 2012;65:329-52. doi: 10.1007/978-94-007-5416-4_14. Subcell Biochem. 2012. PMID: 23225010 Review.
-
Effects of CX3CR1 and Fractalkine Chemokines in Amyloid Beta Clearance and p-Tau Accumulation in Alzheimer's Disease (AD) Rodent Models: Is Fractalkine a Systemic Biomarker for AD?Curr Alzheimer Res. 2016;13(4):403-12. doi: 10.2174/1567205013666151116125714. Curr Alzheimer Res. 2016. PMID: 26567742 Review.
Cited by
-
TRIM11 protects against tauopathies and is down-regulated in Alzheimer's disease.Science. 2023 Jul 28;381(6656):eadd6696. doi: 10.1126/science.add6696. Epub 2023 Jul 28. Science. 2023. PMID: 37499037 Free PMC article.
-
Progression of Seed-Induced Aβ Deposition within the Limbic Connectome.Brain Pathol. 2015 Nov;25(6):743-52. doi: 10.1111/bpa.12252. Epub 2015 Mar 24. Brain Pathol. 2015. PMID: 25677332 Free PMC article.
-
Hippocampal CA2 Lewy pathology is associated with cholinergic degeneration in Parkinson's disease with cognitive decline.Acta Neuropathol Commun. 2019 Apr 25;7(1):61. doi: 10.1186/s40478-019-0717-3. Acta Neuropathol Commun. 2019. PMID: 31023342 Free PMC article.
-
From fix to fit into the autoptic human brains.Eur J Histochem. 2018 Aug 27;62(3):2944. doi: 10.4081/ejh.2018.2944. Eur J Histochem. 2018. PMID: 30173504 Free PMC article.
-
Knockout of receptor for advanced glycation end-products attenuates age-related renal lesions.Aging Cell. 2019 Apr;18(2):e12850. doi: 10.1111/acel.12850. Epub 2019 Feb 22. Aging Cell. 2019. PMID: 30794349 Free PMC article.
References
-
- Ballatore C, Lee VM, Trojanowski JQ. 2007. Tau-mediated neurodegeneration in Alzheimer’s disease and related disorders. Nat Rev Neurosci. 8:663–672 - PubMed
-
- Bataille F, Troppmann S, Klebl F, Rogler G, Stoelcker B, Hofstadter F, Bosserhoff AK, Rummele P. 2006. Multiparameter immunofluorescence on paraffin-embedded tissue sections. Appl Immunohistochem Mol Morphol. 14:225–228 - PubMed
-
- Battifora H, Kopinski M. 1986. The influence of protease digestion and duration of fixation on the immunostaining of keratins: a comparison of formalin and ethanol fixation. J Histochem Cytochem. 34:1095–1100 - PubMed
-
- Behrouz N, Defossez A, Delacourte A, Mazzuca M. 1991. The immunohistochemical evidence of amyloid diffuse deposits as a pathological hallmark in Alzheimer’s disease. J Gerontol. 46:B209–B212 - PubMed
-
- Hardy J, Selkoe DJ. 2002. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science. 297:353–356 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

