Spatial domains of progenitor-like cells and functional complexity of a stem cell niche in the neonatal rat spinal cord
- PMID: 22821702
- PMCID: PMC3426862
- DOI: 10.1002/stem.1175
Spatial domains of progenitor-like cells and functional complexity of a stem cell niche in the neonatal rat spinal cord
Abstract
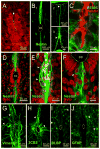
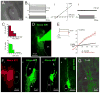
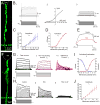
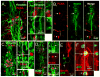
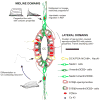
During spinal cord development, progenitors in the neural tube are arranged within spatial domains that generate specific cell types. The ependyma of the postnatal spinal cord seems to retain cells with properties of the primitive neural stem cells, some of which are able to react to injury with active proliferation. However, the functional complexity and organization of this stem cell niche in mammals remains poorly understood. Here, we combined immunohistochemistry for cell-specific markers with patch-clamp recordings to test the hypothesis that the ependyma of the neonatal rat spinal cord contains progenitor-like cells functionally segregated within specific domains. Cells on the lateral aspects of the ependyma combined morphological and molecular traits of ependymocytes and radial glia (RG) expressing S100β and vimentin, displayed passive membrane properties and were electrically coupled via Cx43. Cells contacting the ventral and dorsal poles expressed the neural stem cell markers nestin and/or vimentin, had the typical morphology of RG, and appeared uncoupled displaying various combinations of K(+) and Ca(2+) voltage-gated currents. Although progenitor-like cells were mitotically active around the entire ependyma, the proliferative capacity seemed higher on lateral domains. Our findings represent the first evidence that the ependyma of the rat harbors progenitor-like cells with heterogeneous electrophysiological phenotypes organized in spatial domains. The manipulation of specific functional properties in the heterogeneous population of progenitor-like cells contacting the ependyma may in future help to regulate their behavior and lineage potential, providing the cell types required for the endogenous repair of the injured spinal cord.
Copyright © 2012 AlphaMed Press.
Conflict of interest statement
The authors declare no conflicts of interests.
Figures


References
-
- Ming GL, Song H. Adult neurogenesis in the mammalian central nervous system. Annu Rev Neurosci. 2005;28:223–250. - PubMed
-
- Lledo PM, Alonso M, Grubb MS. Adult neurogenesis and functional plasticity in neuronal circuits. Nat Rev Neurosci. 2006;7:179–193. - PubMed
-
- Hugnot JP, Franzen R. The spinal cord ependymal region: a stem cell niche in the caudal central nervous system. Front Biosci. 2011;16:1044–1059. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

