Modeling the signatures of hydrides in metalloenzymes: ENDOR analysis of a Di-iron Fe(μ-NH)(μ-H)Fe core
- PMID: 22823933
- PMCID: PMC3433054
- DOI: 10.1021/ja303739g
Modeling the signatures of hydrides in metalloenzymes: ENDOR analysis of a Di-iron Fe(μ-NH)(μ-H)Fe core
Abstract
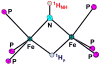
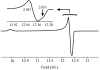
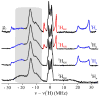
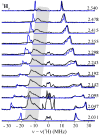
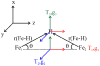
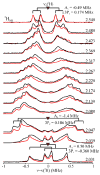
The application of 35 GHz pulsed EPR and ENDOR spectroscopies has established that the biomimetic model complex L(3)Fe(μ-NH)(μ-H)FeL(3) (L(3) = [PhB(CH(2)PPh(2))(3)](-)) complex, 3, is a novel S = (1)/(2) type-III mixed-valence di-iron II/III species, in which the unpaired electron is shared equally between the two iron centers. (1,2)H and (14,15)N ENDOR measurements of the bridging imide are consistent with an allyl radical molecular orbital model for the two bridging ligands. Both the (μ-H) and the proton of the (μ-NH) of the crystallographically characterized 3 show the proposed signature of a 'bridging' hydride that is essentially equidistant between two 'anchor' metal ions: a rhombic dipolar interaction tensor, T ≈ [T, -T, 0]. The point-dipole model for describing the anisotropic interaction of a bridging H as the sum of the point-dipole couplings to the 'anchor' metal ions reproduces this signature with high accuracy, as well as the axial tensor of a terminal hydride, T ≈ [-T, -T, 2T], thus validating both the model and the signatures. This validation in turn lends strong support to the assignment, based on such a point-dipole analysis, that the molybdenum-iron cofactor of nitrogenase contains two [Fe-H(-)-Fe] bridging-hydride fragments in the catalytic intermediate that has accumulated four reducing equivalents (E(4)). Analysis further reveals a complementary similarity between the isotropic hyperfine couplings for the bridging hydrides in 3 and E(4). This study provides a foundation for spectroscopic study of hydrides in a variety of reducing metalloenzymes in addition to nitrogenase.
Figures




References
-
- Igarashi RY, Laryukhin M, Santos PCD, Lee H-I, Dean DR, Seefeldt LC, Hoffman BM. J Am Chem Soc. 2005;127:6231. - PubMed
-
- Fan C, Teixeira M, Moura J, Moura I, Huynh B-H, le Gall J, Peck HD, Jr, Hoffman BM. J Am Chem Soc. 1991;113:20.
-
- Stein M, Lubitz W. Phys Chem Chem Phys. 2001;3:5115.
-
- Lubitz W, Reijerse E, van Gastel M. Chem Rev (Washington, DC, U S) 2007;107:4331. - PubMed
-
- Amara P, Mouesca J-M, Volbeda A, Fontecilla-Camps JC. Inorg Chem (Washington, DC, U S) 2011;50:1868. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

