Hepatic immunosuppressive effects of systemically administered novel dextran-methylprednisolone prodrugs with peptide linkers in rats
- PMID: 22829462
- PMCID: PMC3426655
- DOI: 10.1002/jps.23274
Hepatic immunosuppressive effects of systemically administered novel dextran-methylprednisolone prodrugs with peptide linkers in rats
Abstract
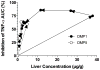
The hepatic immunosuppressive activities of two novel dextran prodrugs of methylprednisolone (MP) containing one (DMP1) or five (DMP5) amino acids as linkers were studied in rats. At various times (0-2 weeks) after intravenous administration of single 5 mg/kg (MP equivalent) doses of each prodrug or MP succinate (MPS), livers were isolated and immunologically stimulated ex vivo with lipopolysaccharide. The concentrations of tumor necrosis factor (TNF)-α in the outlet perfusate were then quantitated to assess immune response. Additionally, the concentrations of DMP1, DMP5, and/or MP were measured in the liver. MPS, DMP5, or DMP1 injections caused a maximum of 48.9%, 63.5%, or 85.7% decrease in the TNF-α secretion into the perfusate, with the time above the 50% inhibitory effect being <5, <24, or 120 h, respectively. Additionally, the area under the effect-time curve for DMP1 was 11-fold or fourfold higher than that after the administration of MPS or DMP5, respectively. Relatively high concentrations of DMP1 were present in the liver even at the last sampling time of 2 weeks. These data suggest that a single intravenous dose of DMP1 produces an intense and sustained immunosuppression in the liver for a relatively long time, which may be useful in liver transplantation.
Copyright © 2012 Wiley Periodicals, Inc.
Figures



Similar articles
-
Dextran-methylprednisolone succinate as a prodrug of methylprednisolone: local immunosuppressive effects in liver after systemic administration to rats.Pharm Res. 2003 Feb;20(2):198-204. doi: 10.1023/a:1022358702643. Pharm Res. 2003. PMID: 12636157
-
Plasma pharmacokinetics and tissue disposition of novel dextran-methylprednisolone conjugates with peptide linkers in rats.J Pharm Sci. 2010 Mar;99(3):1626-37. doi: 10.1002/jps.21934. J Pharm Sci. 2010. PMID: 19780131 Free PMC article.
-
Dextran-methylprednisolone succinate as a prodrug of methylprednisolone: immunosuppressive effects after in vivo administration to rats.Pharm Res. 2000 Nov;17(11):1402-7. doi: 10.1023/a:1007555107691. Pharm Res. 2000. PMID: 11205734
-
Dextran-methylprednisolone succinate as a prodrug of methylprednisolone: in vitro immunosuppressive effects on rat blood and spleen lymphocytes.Int J Pharm. 2000 Oct 10;207(1-2):71-6. doi: 10.1016/s0378-5173(00)00544-5. Int J Pharm. 2000. PMID: 11036232
-
Methylprednisolone suleptanate Pharmacia Corp.Curr Opin Investig Drugs. 2000 Sep;1(1):97-103. Curr Opin Investig Drugs. 2000. PMID: 11249603 Review.
References
-
- Encke J, Uhl W, Stremmel W, Sauer P. Immunosuppression and modulation in liver transplantation. Nephrol Dial Transplant. 2004;19(Suppl 4):IV22–IV25. - PubMed
-
- Volpin R, Angeli P, Galioto A, Fasolato S, Neri D, Barbazza F, Merenda R, Del Piccolo F, Strazzabosco M, Casagrande F, Feltracco P, Sticca A, Merkel C, Gerunda G, Gatta A. Comparison between two high-dose methylprednisolone schedules in the treatment of acute hepatic cellular rejection in liver transplant recipients: a controlled clinical trial. Liver Transpl. 2002;8:527–534. - PubMed
-
- Yu S, Wu L, Jin J, Yan S, Jiang G, Xie H, Zheng S. Influence of CYP3A5 gene polymorphisms of donor rather than recipient to tacrolimus individual dose requirement in liver transplantation. Transplantation. 2006;81:46–51. - PubMed
-
- Wiesner R, Ludwig J, Krom R, Steers J, Porayko M, Gores G, Hay J. Treatment of early cellular rejection following liver transplantation with intravenous methylprednisolone. The effect of dose on response. Transplantation. 1994;58:1053–1056. - PubMed
-
- Reyes J, Todo S, Starzl TE. Liver and intestine transplantation. Immunol Allergy Clin North Am. 1996;16:293.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

