Reconstitution of the central and peripheral nervous system during salamander tail regeneration
- PMID: 22829665
- PMCID: PMC3427070
- DOI: 10.1073/pnas.1116738109
Reconstitution of the central and peripheral nervous system during salamander tail regeneration
Abstract
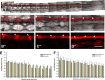
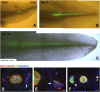
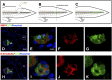
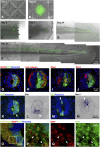
We show that after tail amputation in Ambystoma mexicanum (Axolotl) the correct number and spacing of dorsal root ganglia are regenerated. By transplantation of spinal cord tissue and nonclonal neurospheres, we show that the central spinal cord represents a source of peripheral nervous system cells. Interestingly, melanophores migrate from preexisting precursors in the skin. Finally, we demonstrate that implantation of a clonally derived spinal cord neurosphere can result in reconstitution of all examined cell types in the regenerating central spinal cord, suggesting derivation of a cell with spinal cord stem cell properties.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Iten LE, Bryant SV. Regeneration from different levels along the tail of the newt, Notophthalmus viridescens. J Exp Zool. 1976;196:293–306. - PubMed
-
- Arsanto JP, et al. Formation of the peripheral nervous system during tail regeneration in urodele amphibians: Ultrastructural and immunohistochemical studies of the origin of the cells. J Exp Zool. 1992;264:273–292. - PubMed
-
- Benraiss A, Arsanto JP, Coulon J, Thouveny Y. Neural crest-like cells originate from the spinal cord during tail regeneration in adult amphibian urodeles. Dev Dyn. 1997;209:15–28. - PubMed
-
- Géraudie J, Nordlander R, Singer M, Singer J. Early stages of spinal ganglion formation during tail regeneration in the newt, Notophthalmus viridescens. Am J Anat. 1988;183:359–370. - PubMed
-
- Echeverri K, Tanaka EM. Ectoderm to mesoderm lineage switching during axolotl tail regeneration. Science. 2002;298:1993–1996. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

