Long telomeres bypass the requirement for telomere maintenance in human tumorigenesis
- PMID: 22832159
- PMCID: PMC3406325
- DOI: 10.1016/j.celrep.2011.12.004
Long telomeres bypass the requirement for telomere maintenance in human tumorigenesis
Abstract
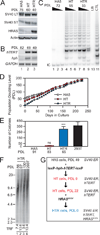
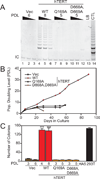
Despite the importance of telomere maintenance in cancer cell survival via the elongation of telomeres by telomerase reverse transcriptase (TERT) or alternative lengthening of telomeres (ALT), it had not been tested directly whether telomere maintenance is dispensable for human tumorigenesis. We engineered human tumor cells containing loxP-flanked hTERT to enable extensive telomere elongation prior to complete hTERT excision. Despite unabated telomere erosion, hTERT-excised cells formed tumors in mice and proliferated in vitro for up to 1 year. Telomerase reactivation or ALT was not observed, and the eventual loss of telomeric signal coincided with loss of tumorigenic potential and cell viability. Crisis was averted via the reintroduction of active but not inactive hTERT. Thus, telomere maintenance is dispensable for human tumorigenesis when telomere reserves are long. Yet, despite telomere instability and the presence of oncogenic RAS, human tumors remain susceptible to crisis induced by critically short telomeres.
Copyright © 2012 The Authors. Published by Elsevier Inc. All rights reserved.
Figures


Similar articles
-
Immortal ALT+ human cells do not require telomerase reverse transcriptase for malignant transformation.Cancer Res. 2005 Aug 1;65(15):6512-5. doi: 10.1158/0008-5472.CAN-05-1210. Cancer Res. 2005. PMID: 16061628
-
Super-telomeres in transformed human fibroblasts.Biochim Biophys Acta. 2013 Aug;1833(8):1885-93. doi: 10.1016/j.bbamcr.2013.03.030. Epub 2013 Apr 6. Biochim Biophys Acta. 2013. PMID: 23570868
-
Telomerase- and alternative telomere lengthening-independent telomere stabilization in a metastasis-derived human non-small cell lung cancer cell line: effect of ectopic hTERT.Cancer Res. 2006 Apr 1;66(7):3584-92. doi: 10.1158/0008-5472.CAN-05-2839. Cancer Res. 2006. PMID: 16585183
-
Alternative mechanisms of telomere lengthening: permissive mutations, DNA repair proteins and tumorigenic progression.Mutat Res. 2013 Mar-Apr;743-744:142-150. doi: 10.1016/j.mrfmmm.2012.11.006. Epub 2012 Dec 3. Mutat Res. 2013. PMID: 23219603 Free PMC article. Review.
-
Telomere Length Maintenance in Cancer: At the Crossroad between Telomerase and Alternative Lengthening of Telomeres (ALT).Int J Mol Sci. 2018 Feb 18;19(2):606. doi: 10.3390/ijms19020606. Int J Mol Sci. 2018. PMID: 29463031 Free PMC article. Review.
Cited by
-
Rare missense variants in POT1 predispose to familial cutaneous malignant melanoma.Nat Genet. 2014 May;46(5):482-6. doi: 10.1038/ng.2941. Epub 2014 Mar 30. Nat Genet. 2014. PMID: 24686846 Free PMC article.
-
Dissecting Fission Yeast Shelterin Interactions via MICro-MS Links Disruption of Shelterin Bridge to Tumorigenesis.Cell Rep. 2015 Sep 29;12(12):2169-80. doi: 10.1016/j.celrep.2015.08.043. Epub 2015 Sep 10. Cell Rep. 2015. PMID: 26365187 Free PMC article.
-
TINF2 is a haploinsufficient tumor suppressor that limits telomere length.Elife. 2020 Dec 1;9:e61235. doi: 10.7554/eLife.61235. Elife. 2020. PMID: 33258446 Free PMC article.
-
A yeast chemical genetic screen identifies inhibitors of human telomerase.Chem Biol. 2013 Mar 21;20(3):333-40. doi: 10.1016/j.chembiol.2012.12.008. Chem Biol. 2013. PMID: 23521791 Free PMC article.
-
Telomerase-Mediated Anti-Ageing Interventions.Subcell Biochem. 2024;107:1-20. doi: 10.1007/978-3-031-66768-8_1. Subcell Biochem. 2024. PMID: 39693017 Review.
References
-
- Bodnar AG, Ouellette M, Frolkis M, Holt SE, Chiu CP, Morin GB, Harley CB, Shay JW, Lichtsteiner S, Wright WE. Extension of life-span by introduction of telomerase into normal human cells. Science. 1998;279:349–352. - PubMed
-
- Cascio SM. Novel strategies for immortalization of human hepatocytes. Artif. Organs. 2001;25:529–538. - PubMed
-
- Cesare AJ, Reddel RR. Alternative lengthening of telomeres: models, mechanisms and implications. Nat. Rev. Genet. 2010;11:319–330. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

