Neural circuitry engaged by prostaglandins during the sickness syndrome
- PMID: 22837039
- PMCID: PMC3748818
- DOI: 10.1038/nn.3159
Neural circuitry engaged by prostaglandins during the sickness syndrome
Abstract
During illnesses caused by infectious disease or other sources of inflammation, a suite of brain-mediated responses called the sickness syndrome occurs, which includes fever, anorexia, sleepiness, hyperalgesia and elevated corticosteroid secretion. Much of the sickness syndrome is mediated by prostaglandins acting on the brain and can be prevented by nonsteroidal anti-inflammatory drugs, such as aspirin or ibuprofen, that block prostaglandin synthesis. By examining which prostaglandins are produced at which sites and how they interact with the nervous system, researchers have identified specific neural circuits that underlie the sickness syndrome.
Figures
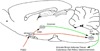

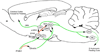
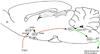
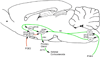
References
-
- Kozak W, Conn CA, Kluger MJ. Lipopolysaccharide induces fever and depresses locomotor activity in unrestrained mice. Am. J. Physiol. 1994;266:R125–R135. - PubMed
-
- Elmquist JK, Scammell TE, Saper CB. Mechanisms of CNS response to systemic immune challenge: the febrile response. Trends Neurosci. 1997;20:565–570. - PubMed
-
- Konsman JP, Parnet P, Dantzer R. Cytokine-induced sickness behaviour: mechanisms and implications. Trends Neurosci. 2002;25:154–159. - PubMed
-
- Romanovsky AA, et al. First and second phases of biphasic fever: two sequential stages of the sickness syndrome? Am. J. Physiol. 1996;271:R244–R253. - PubMed
-
- Kluger MJ, Kozak W, Conn CA, Leon LR, Soszynski D. Role of fever in disease. Ann. N. Y. Acad. Sci. 1998;856:224–233. 224–233. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

