Rapamycin reverses elevated mTORC1 signaling in lamin A/C-deficient mice, rescues cardiac and skeletal muscle function, and extends survival
- PMID: 22837538
- PMCID: PMC3613228
- DOI: 10.1126/scitranslmed.3003802
Rapamycin reverses elevated mTORC1 signaling in lamin A/C-deficient mice, rescues cardiac and skeletal muscle function, and extends survival
Abstract
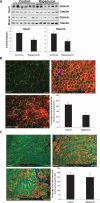
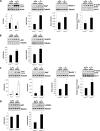
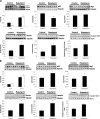
Mutations in LMNA, the gene that encodes A-type lamins, cause multiple diseases including dystrophies of the skeletal muscle and fat, dilated cardiomyopathy, and progeria-like syndromes (collectively termed laminopathies). Reduced A-type lamin function, however, is most commonly associated with skeletal muscle dystrophy and dilated cardiomyopathy rather than lipodystrophy or progeria. The mechanisms underlying these diseases are only beginning to be unraveled. We report that mice deficient in Lmna, which corresponds to the human gene LMNA, have enhanced mTORC1 (mammalian target of rapamycin complex 1) signaling specifically in tissues linked to pathology, namely, cardiac and skeletal muscle. Pharmacologic reversal of elevated mTORC1 signaling by rapamycin improves cardiac and skeletal muscle function and enhances survival in mice lacking A-type lamins. At the cellular level, rapamycin decreases the number of myocytes with abnormal desmin accumulation and decreases the amount of desmin in both muscle and cardiac tissue of Lmna(-/-) mice. In addition, inhibition of mTORC1 signaling with rapamycin improves defective autophagic-mediated degradation in Lmna(-/-) mice. Together, these findings point to aberrant mTORC1 signaling as a mechanistic component of laminopathies associated with reduced A-type lamin function and offer a potential therapeutic approach, namely, the use of rapamycin-related mTORC1 inhibitors.
Figures




References
-
- Taimen P, Pfleghaar K, Shimi T, Möller D, Ben-Harush K, Erdos MR, Adam SA, Herrmann H, Medalia O, Collins FS, Goldman AE, Goldman RD. A progeria mutation reveals functions for lamin A in nuclear assembly, architecture, and chromosome organization. Proc. Natl. Acad. Sci. U.S.A. 2009;106:20788–20793. - PMC - PubMed
-
- Lattanzi G, Columbaro M, Mattioli E, Cenni V, Camozzi D, Wehnert M, Santi S, Riccio M, Del Coco R, Maraldi NM, Squarzoni S, Foisner R, Capanni C. Pre-lamin A processing is linked to heterochromatin organization. J. Cell. Biochem. 2007;102:1149–1159. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

