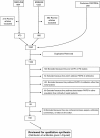
Does Beta-lactam Pharmacokinetic Variability in Critically Ill Patients Justify Therapeutic Drug Monitoring? A Systematic Review
- PMID: 22839761
- PMCID: PMC3460787
- DOI: 10.1186/2110-5820-2-35
Does Beta-lactam Pharmacokinetic Variability in Critically Ill Patients Justify Therapeutic Drug Monitoring? A Systematic Review
Abstract
The pharmacokinetics of beta-lactam antibiotics in intensive care patients may be profoundly altered due to the dynamic, unpredictable pathophysiological changes that occur in critical illness. For many drugs, significant increases in the volume of distribution and/or variability in drug clearance are common. When "standard" beta-lactam doses are used, such pharmacokinetic changes can result in subtherapeutic plasma concentrations, treatment failure, and the development of antibiotic resistance. Emerging data support the use of beta-lactam therapeutic drug monitoring (TDM) and individualized dosing to ensure the achievement of pharmacodynamic targets associated with rapid bacterial killing and optimal clinical outcomes. The purpose of this work was to describe the pharmacokinetic variability of beta-lactams in the critically ill and to discuss the potential utility of TDM to optimize antibiotic therapy through a structured literature review of all relevant publications between 1946 and October 2011. Only a few studies have reported the utility of TDM as a tool to improve beta-lactam dosing in critically ill patients. Moreover, there is little agreement between studies on the pharmacodynamic targets required to optimize antibiotic therapy. The impact of TDM on important clinical outcomes also remains to be established. Whereas TDM may be theoretically rational, clinical studies to assess utility in the clinical setting are urgently required.
Figures
References
-
- Ariano RE, Nyhlen A, Donnelly JP, Sitar DS, Harding GK, Zelenitsky SA. Pharmacokinetics and pharmacodynamics of meropenem in febrile neutropenic patients with bacteremia. Ann Pharmacother. 2005;39:32–38. - PubMed
LinkOut - more resources
Full Text Sources
Medical