Nanomolar ouabain increases NCX1 expression and enhances Ca2+ signaling in human arterial myocytes: a mechanism that links salt to increased vascular resistance?
- PMID: 22842068
- PMCID: PMC3469703
- DOI: 10.1152/ajpheart.00399.2012
Nanomolar ouabain increases NCX1 expression and enhances Ca2+ signaling in human arterial myocytes: a mechanism that links salt to increased vascular resistance?
Abstract
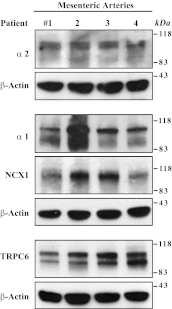
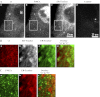
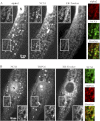
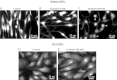
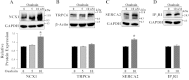
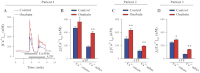
The mechanisms by which NaCl raises blood pressure (BP) in hypertension are unresolved, but much evidence indicates that endogenous ouabain is involved. In rodents, arterial smooth muscle cell (ASMC) Na(+) pumps with an α(2)-catalytic subunit (ouabain EC(50) ≤1.0 nM) are crucial for some hypertension models, even though ≈80% of ASMC Na(+) pumps have an α(1)-subunit (ouabain EC(50) ≈ 5 μM). Human α(1)-Na(+) pumps, however, have high ouabain affinity (EC(50) ≈ 10-20 nM). We used immunoblotting, immunocytochemistry, and Ca(2+) imaging (fura-2) to examine the expression, distribution, and function of Na(+) pump α-subunit isoforms in human arteries and primary cultured human ASMCs (hASMCs). hASMCs express α(1)- and α(2)-Na(+) pumps. Further, α(2)-, but not α(1)-, pumps are confined to plasma membrane microdomains adjacent to sarcoplasmic reticulum (SR), where they colocalize with Na/Ca exchanger-1 (NCX1) and C-type transient receptor potential-6 (receptor-operated channels, ROCs). Prolonged inhibition (72 h) with 100 nM ouabain (blocks nearly all α(1)- and α(2)-pumps) was toxic to most cultured hASMCs. Treatment with 10 nM ouabain (72 h), however, increased NCX1 and sarco(endo)plasmic reticulum Ca(2+)-ATPase expression and augmented ATP (10 μM)-induced SR Ca(2+) release in 0 Ca(2+), ouabain-free media, and Ca(2+) influx after external Ca(2+) restoration. The latter was likely mediated primarily by ROCs and store-operated Ca(2+) channels. These hASMC protein expression and Ca(2+) signaling changes are comparable with previous observations on myocytes isolated from arteries of many rat hypertension models. We conclude that the same structurally and functionally coupled mechanisms (α(2)-Na(+) pumps, NCX1, ROCs, and the SR) regulate Ca(2+) homeostasis and signaling in hASMCs and rodent ASMCs. These ouabain/endogenous ouabain-modulated mechanisms underlie the whole body autoregulation associated with increased vascular resistance and elevation of BP in human, salt-sensitive hypertension.
Figures

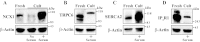

Similar articles
-
Arterial α2-Na+ pump expression influences blood pressure: lessons from novel, genetically engineered smooth muscle-specific α2 mice.Am J Physiol Heart Circ Physiol. 2015 Sep;309(5):H958-68. doi: 10.1152/ajpheart.00430.2015. Epub 2015 Jul 24. Am J Physiol Heart Circ Physiol. 2015. PMID: 26209057 Free PMC article.
-
Upregulation of Na+ and Ca2+ transporters in arterial smooth muscle from ouabain-induced hypertensive rats.Am J Physiol Heart Circ Physiol. 2010 Jan;298(1):H263-74. doi: 10.1152/ajpheart.00784.2009. Epub 2009 Nov 6. Am J Physiol Heart Circ Physiol. 2010. PMID: 19897708 Free PMC article.
-
Activation of c-SRC underlies the differential effects of ouabain and digoxin on Ca(2+) signaling in arterial smooth muscle cells.Am J Physiol Cell Physiol. 2013 Feb 15;304(4):C324-33. doi: 10.1152/ajpcell.00337.2012. Epub 2012 Nov 28. Am J Physiol Cell Physiol. 2013. PMID: 23195071 Free PMC article.
-
Signaling mechanisms that link salt retention to hypertension: endogenous ouabain, the Na(+) pump, the Na(+)/Ca(2+) exchanger and TRPC proteins.Biochim Biophys Acta. 2010 Dec;1802(12):1219-29. doi: 10.1016/j.bbadis.2010.02.011. Epub 2010 Mar 6. Biochim Biophys Acta. 2010. PMID: 20211726 Free PMC article. Review.
-
How does salt retention raise blood pressure?Am J Physiol Regul Integr Comp Physiol. 2006 Mar;290(3):R514-23. doi: 10.1152/ajpregu.00819.2005. Am J Physiol Regul Integr Comp Physiol. 2006. PMID: 16467498 Review.
Cited by
-
How does pressure overload cause cardiac hypertrophy and dysfunction? High-ouabain affinity cardiac Na+ pumps are crucial.Am J Physiol Heart Circ Physiol. 2017 Nov 1;313(5):H919-H930. doi: 10.1152/ajpheart.00131.2017. Epub 2017 Jul 21. Am J Physiol Heart Circ Physiol. 2017. PMID: 28733446 Free PMC article. Review.
-
Ouabain-digoxin antagonism in rat arteries and neurones.J Physiol. 2014 Mar 1;592(5):941-69. doi: 10.1113/jphysiol.2013.266866. Epub 2013 Dec 16. J Physiol. 2014. PMID: 24344167 Free PMC article.
-
The vascular Na,K-ATPase: clinical implications in stroke, migraine, and hypertension.Clin Sci (Lond). 2023 Oct 31;137(20):1595-1618. doi: 10.1042/CS20220796. Clin Sci (Lond). 2023. PMID: 37877226 Free PMC article. Review.
-
Endogenous cardiotonic steroids in kidney failure: a review and an hypothesis.Adv Chronic Kidney Dis. 2015 May;22(3):232-44. doi: 10.1053/j.ackd.2014.12.005. Adv Chronic Kidney Dis. 2015. PMID: 25908473 Free PMC article. Review.
-
Arterial α2-Na+ pump expression influences blood pressure: lessons from novel, genetically engineered smooth muscle-specific α2 mice.Am J Physiol Heart Circ Physiol. 2015 Sep;309(5):H958-68. doi: 10.1152/ajpheart.00430.2015. Epub 2015 Jul 24. Am J Physiol Heart Circ Physiol. 2015. PMID: 26209057 Free PMC article.
References
-
- Bae YM, Kim A, Lee YJ, Lim W, Noh YH, Kim EJ, Kim J, Kim TK, Park SW, Kim B, Cho SI, Kim DK, Ho WK. Enhancement of receptor-operated cation current and TRPC6 expression in arterial smooth muscle cells of deoxycorticosterone acetate-salt hypertensive rats. J Hypertens 25: 809–817, 2007 - PubMed
-
- Baryshnikov SG, Pulina MV, Zulian A, Linde CI, Golovina VA. Orai1, a critical component of store-operated Ca2+ entry, is functionally associated with Na+/Ca2+ exchanger and plasma membrane Ca2+ pump in proliferating human arterial myocytes. Am J Physiol Cell Physiol 297: C1103–C1112, 2009 - PMC - PubMed
-
- Berra-Romani R, Blaustein MP, Matteson DR. TTX-sensitive voltage-gated Na+ channels are expressed in mesenteric artery smooth muscle cells. Am J Physiol Heart Circ Physiol 289: H137–H145, 2005 - PubMed
-
- Blanco G, Mercer RW. Isozymes of the Na-K-ATPase: heterogeneity in structure, diversity in function. Am J Physiol Renal Physiol 275: F633–F650, 1998 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

