Exploring the role of a conserved class A residue in the Ω-Loop of KPC-2 β-lactamase: a mechanism for ceftazidime hydrolysis
- PMID: 22843686
- PMCID: PMC3442512
- DOI: 10.1074/jbc.M112.348540
Exploring the role of a conserved class A residue in the Ω-Loop of KPC-2 β-lactamase: a mechanism for ceftazidime hydrolysis
Abstract
Gram-negative bacteria harboring KPC-2, a class A β-lactamase, are resistant to all β-lactam antibiotics and pose a major public health threat. Arg-164 is a conserved residue in all class A β-lactamases and is located in the solvent-exposed Ω-loop of KPC-2. To probe the role of this amino acid in KPC-2, we performed site-saturation mutagenesis. When compared with wild type, 11 of 19 variants at position Arg-164 in KPC-2 conferred increased resistance to the oxyimino-cephalosporin, ceftazidime (minimum inhibitory concentration; 32→128 mg/liter) when expressed in Escherichia coli. Using the R164S variant of KPC-2 as a representative β-lactamase for more detailed analysis, we observed only a modest 25% increase in k(cat)/K(m) for ceftazidime (0.015→0.019 μm(-1) s(-1)). Employing pre-steady-state kinetics and mass spectrometry, we determined that acylation is rate-limiting for ceftazidime hydrolysis by KPC-2, whereas deacylation is rate-limiting in the R164S variant, leading to accumulation of acyl-enzyme at steady-state. CD spectroscopy revealed that a conformational change occurred in the turnover of ceftazidime by KPC-2, but not the R164S variant, providing evidence for a different form of the enzyme at steady state. Molecular models constructed to explain these findings suggest that ceftazidime adopts a unique conformation, despite preservation of Ω-loop structure. We propose that the R164S substitution in KPC-2 enhances ceftazidime resistance by proceeding through "covalent trapping" of the substrate by a deacylation impaired enzyme with a lower K(m). Future antibiotic design must consider the distinctive behavior of the Ω-loop of KPC-2.
Figures


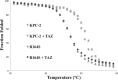
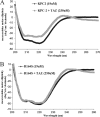

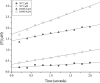
Comment in
-
Covalent trapping and bacterial resistance to ceftazidime.J Biol Chem. 2013 Sep 13;288(37):26967. doi: 10.1074/jbc.L113.493676. J Biol Chem. 2013. PMID: 24037546 Free PMC article. No abstract available.
-
Reply to Frère: Covalent trapping and bacterial resistance to ceftazidime.J Biol Chem. 2013 Sep 13;288(37):26968. doi: 10.1074/jbc.l113.502237. J Biol Chem. 2013. PMID: 24163830 Free PMC article. No abstract available.
References
-
- Bush K., Fisher J. F. (2011) Epidemiological expansion, structural studies, and clinical challenges of new β-lactamases from Gram-negative bacteria. Annu. Rev. Microbiol. 65, 455–478 - PubMed
-
- Barlow M. (2009) What antimicrobial resistance has taught us about horizontal gene transfer. Methods Mol. Biol. 532, 397–411 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

