Neuroprotective effects of L-carnitine against oxygen-glucose deprivation in rat primary cortical neurons
- PMID: 22844318
- PMCID: PMC3405156
- DOI: 10.3345/kjp.2012.55.7.238
Neuroprotective effects of L-carnitine against oxygen-glucose deprivation in rat primary cortical neurons
Abstract
Purpose: Hypoxic-ischemic encephalopathy is an important cause of neonatal mortality, as this brain injury disrupts normal mitochondrial respiratory activity. Carnitine plays an essential role in mitochondrial fatty acid transport and modulates excess acyl coenzyme A levels. In this study, we investigated whether treatment of primary cultures of rat cortical neurons with L-carnitine was able to prevent neurotoxicity resulting from oxygen-glucose deprivation (OGD).
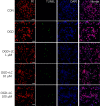
Methods: Cortical neurons were prepared from Sprague-Dawley rat embryos. L-Carnitine was applied to cultures just prior to OGD and subsequent reoxygenation. The numbers of cells that stained with acridine orange (AO) and propidium iodide (PI) were counted, and lactate dehydrogenase (LDH) activity and reactive oxygen species (ROS) levels were measured. The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay and the terminal uridine deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling assay were performed to evaluate the effect of L-carnitine (1 µM, 10 µM, and 100 µM) on OGD-induced neurotoxicity.
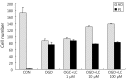
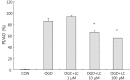
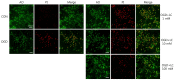
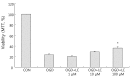
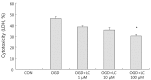
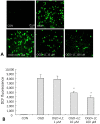
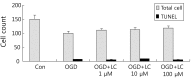
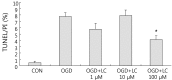
Results: Treatment of primary cultures of rat cortical neurons with L-carnitine significantly reduced cell necrosis and prevented apoptosis after OGD. L-Carnitine application significantly reduced the number of cells that died, as assessed by the PI/AO ratio, and also reduced ROS release in the OGD groups treated with 10 µM and 100 µM of L-carnitine compared with the untreated OGD group (P<0.05). The application of L-carnitine at 100 µM significantly decreased cytotoxicity, LDH release, and inhibited apoptosis compared to the untreated OGD group (P<0.05).
Conclusion: L-Carnitine has neuroprotective benefits against OGD in rat primary cortical neurons in vitro.
Keywords: Hypoxia-Ischemia; L-carnitine; Neurons; Neuroprotective effect; Oxygen-glucose deprivation.
Figures
Similar articles
-
Neuroprotective effects of orientin on oxygen-glucose deprivation/reperfusion-induced cell injury in primary culture of rat cortical neurons.Exp Biol Med (Maywood). 2018 Jan;243(1):78-86. doi: 10.1177/1535370217737983. Epub 2017 Oct 26. Exp Biol Med (Maywood). 2018. PMID: 29073777 Free PMC article.
-
Neuroprotective effect of licochalcone A against oxygen-glucose deprivation/reperfusion in rat primary cortical neurons by attenuating oxidative stress injury and inflammatory response via the SIRT1/Nrf2 pathway.J Cell Biochem. 2018 Apr;119(4):3210-3219. doi: 10.1002/jcb.26477. Epub 2017 Dec 26. J Cell Biochem. 2018. PMID: 29105819
-
Anthocyanins extracted from black soybean seed coat protect primary cortical neurons against in vitro ischemia.Biol Pharm Bull. 2012;35(7):999-1008. doi: 10.1248/bpb.b110628. Biol Pharm Bull. 2012. PMID: 22791144
-
The neuroprotective role and mechanisms of TERT in neurons with oxygen-glucose deprivation.Neuroscience. 2013 Nov 12;252:346-58. doi: 10.1016/j.neuroscience.2013.08.015. Epub 2013 Aug 19. Neuroscience. 2013. PMID: 23968592
-
L-carnitine protects neurons from 1-methyl-4-phenylpyridinium-induced neuronal apoptosis in rat forebrain culture.Neuroscience. 2007 Jan 5;144(1):46-55. doi: 10.1016/j.neuroscience.2006.08.083. Epub 2006 Nov 2. Neuroscience. 2007. PMID: 17084538
Cited by
-
Metabolic Response of Pediatric Traumatic Brain Injury.J Child Neurol. 2016 Jan;31(1):28-34. doi: 10.1177/0883073814549244. Epub 2014 Oct 21. J Child Neurol. 2016. PMID: 25336427 Free PMC article. Review.
-
Thrombin Preconditioning Enhances Therapeutic Efficacy of Human Wharton's Jelly-Derived Mesenchymal Stem Cells in Severe Neonatal Hypoxic Ischemic Encephalopathy.Int J Mol Sci. 2019 May 20;20(10):2477. doi: 10.3390/ijms20102477. Int J Mol Sci. 2019. PMID: 31137455 Free PMC article.
-
L-carnitine contributes to enhancement of neurogenesis from mesenchymal stem cells through Wnt/β-catenin and PKA pathway.Exp Biol Med (Maywood). 2017 Mar;242(5):482-486. doi: 10.1177/1535370216685432. Epub 2017 Jan 5. Exp Biol Med (Maywood). 2017. PMID: 28056548 Free PMC article.
-
Targeting Alzheimer's disease with multimodal polypeptide-based nanoconjugates.Sci Adv. 2021 Mar 26;7(13):eabf9180. doi: 10.1126/sciadv.abf9180. Print 2021 Mar. Sci Adv. 2021. PMID: 33771874 Free PMC article.
-
BDNF-Overexpressing Engineered Mesenchymal Stem Cells Enhances Their Therapeutic Efficacy against Severe Neonatal Hypoxic Ischemic Brain Injury.Int J Mol Sci. 2021 Oct 22;22(21):11395. doi: 10.3390/ijms222111395. Int J Mol Sci. 2021. PMID: 34768827 Free PMC article.
References
-
- Johnston MV, Trescher WH, Ishida A, Nakajima W. Neurobiology of hypoxic-ischemic injury in the developing brain. Pediatr Res. 2001;49:735–741. - PubMed
-
- Vannucci RC. Hypoxic-ischemic encephalopathy. Am J Perinatol. 2000;17:113–120. - PubMed
-
- Aliev G, Smith MA, Obrenovich ME, de la Torre JC, Perry G. Role of vascular hypoperfusion-induced oxidative stress and mitochondria failure in the pathogenesis of Azheimer disease. Neurotox Res. 2003;5:491–504. - PubMed
-
- Gilland E, Puka-Sundvall M, Hillered L, Hagberg H. Mitochondrial function and energy metabolism after hypoxia-ischemia in the immature rat brain: involvement of NMDA-receptors. J Cereb Blood Flow Metab. 1998;18:297–304. - PubMed
-
- Gustavsson M, Anderson MF, Mallard C, Hagberg H. Hypoxic preconditioning confers long-term reduction of brain injury and improvement of neurological ability in immature rats. Pediatr Res. 2005;57:305–309. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

