Chiral Zn(II)-bisamidine complex as a Lewis-Brønsted combined acid catalyst: application to asymmetric Mukaiyama aldol reactions of α-ketoesters
- PMID: 22847141
- PMCID: PMC6268631
- DOI: 10.3390/molecules17089010
Chiral Zn(II)-bisamidine complex as a Lewis-Brønsted combined acid catalyst: application to asymmetric Mukaiyama aldol reactions of α-ketoesters
Abstract
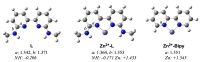
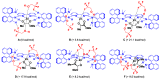
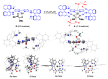
Focusing on the steric and electronic properties of the resonance-stabilized amidine framework, a cationic metal-bisamidine complex was designed as a conjugated combined Lewis-Brønsted acid catalyst. The chiral Zn(II)-bisamidine catalyst prepared from the 2,2'-bipyridyl derived bisamidine ligand, ZnCl₂, and AgSbF₆ promoted asymmetric Mukaiyama aldol reaction of α-ketoester and α,α-disubstituted silyl enol ether to afford the α-hydroxyester having sequential quarternary carbons in good yield, albeit with low enantioselectivity. Addition of 1.0 equivalent of the fluoroalcohol having suitable acidity and bulkiness dramatically increased the enantioselectivity (up to 68% ee). DFT calculations suggested that this additive effect would be caused by self-assembly of the fluoroalcohol on the Zn(II)-bisamidine catalyst.
Figures

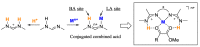

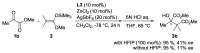
Similar articles
-
Diastereoselectivity in Lewis-acid-catalyzed Mukaiyama aldol reactions: a DFT study.J Am Chem Soc. 2012 Sep 12;134(36):14973-81. doi: 10.1021/ja3052975. Epub 2012 Aug 31. J Am Chem Soc. 2012. PMID: 22891640 Free PMC article.
-
Redox and Lewis acid relay catalysis: a titanocene/zinc catalytic platform in the development of multicomponent coupling reactions.J Org Chem. 2014 Dec 19;79(24):12083-95. doi: 10.1021/jo501890z. Epub 2014 Oct 23. J Org Chem. 2014. PMID: 25279685
-
Super Brønsted acid catalysis.Chem Commun (Camb). 2011 Mar 21;47(11):3043-56. doi: 10.1039/c0cc04867d. Epub 2011 Jan 18. Chem Commun (Camb). 2011. PMID: 21246123
-
The discovery of novel reactivity in the development of C-C bond-forming reactions: in situ generation of zinc acetylides with Zn(II)/R(3)N.Acc Chem Res. 2000 Jun;33(6):373-81. doi: 10.1021/ar990078o. Acc Chem Res. 2000. PMID: 10891055 Review.
-
Asymmetric catalysis of aldol reactions with chiral lewis bases.Acc Chem Res. 2000 Jun;33(6):432-40. doi: 10.1021/ar960027g. Acc Chem Res. 2000. PMID: 10891061 Review.
Cited by
-
One-step multicomponent synthesis of chiral oxazolinyl-zinc complexes.Chem Cent J. 2017 Aug 9;11(1):81. doi: 10.1186/s13065-017-0305-1. Chem Cent J. 2017. PMID: 29086857 Free PMC article.
References
-
- Maruoka K., Sakurai M., Fujiwara J., Yamamoto H. Asymmetric Diels-Alder reaction directed toward chiral anthracycline intermediates. Tetrahedron. 1986;27:4895–4898. doi: 10.1016/S0040-4039(00)85091-1. - DOI
-
- Ishihara K., Miyata M., Hattori K., Tada T., Yamamoto H.Y. A new chiral BLA promoter for asymmetric aza Diels-Alder and Aldol-type reactions of imines. J. Am. Chem. Soc. 1994;116:10520–10524.
-
- Ishihara K., Yamamoto H. Brønsted acid assisted chiral Lewis acid (BLA) catalyst for asymmetric Diels-Alder reaction. J. Am. Chem. Soc. 1994;116:1561–1562. doi: 10.1021/ja00083a048. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

