A possible cross-talk between autophagy and apoptosis in generating an immune response in melanoma
- PMID: 22847295
- PMCID: PMC3747633
- DOI: 10.1007/s10495-012-0745-y
A possible cross-talk between autophagy and apoptosis in generating an immune response in melanoma
Abstract
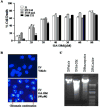
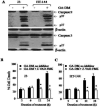
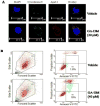
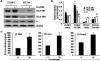
Melanoma is the most aggressive form of skin cancer, responsible for the majority of skin cancer related deaths. Thus, the search for natural molecules which can effectively destroy tumors while promoting immune activation is essential for designing novel therapies against metastatic melanoma. Here, we report for the first time that a natural triterpenoid, Ganoderic acid DM (GA-DM), induces an orchestrated autophagic and apoptotic cell death, as well as enhanced immunological responses via increased HLA class II presentation in melanoma cells. Annexin V staining and flow cytometry showed that GA-DM treatment induced apoptosis of melanoma cells, which was supported by a detection of increased Bax proteins, co-localization and elevation of Apaf-1 and cytochrome c, and a subsequent cleavage of caspases 9 and 3. Furthermore, GA-DM treatment initiated a possible cross-talk between autophagy and apoptosis as evidenced by increased levels of Beclin-1 and LC3 proteins, and their timely interplay with apoptotic and/or anti-apoptotic molecules in melanoma cells. Despite GA-DM's moderate cytotoxicity, viable cells expressed high levels of HLA class II proteins with improved antigen presentation and CD4+ T cell recognition. The antitumor efficacy of GA-DM was also investigated in vivo in murine B16 melanoma model, where GA-DM treatment slowed tumor formation with a significant reduction in tumor volume. Taken together, these findings demonstrate the potential of GA-DM as a natural chemo-immunotherapeutic capable of inducing a possible cross-talk between autophagy and apoptosis, as well as improved immune recognition for sustained melanoma tumor clearance.
Conflict of interest statement
Figures


Similar articles
-
Endoplasmic reticulum stress, autophagic and apoptotic cell death, and immune activation by a natural triterpenoid in human prostate cancer cells.J Cell Biochem. 2019 Apr;120(4):6264-6276. doi: 10.1002/jcb.27913. Epub 2018 Oct 30. J Cell Biochem. 2019. PMID: 30378157 Free PMC article.
-
Ganoderic acid DM induces autophagic apoptosis in non-small cell lung cancer cells by inhibiting the PI3K/Akt/mTOR activity.Chem Biol Interact. 2020 Jan 25;316:108932. doi: 10.1016/j.cbi.2019.108932. Epub 2019 Dec 23. Chem Biol Interact. 2020. PMID: 31874162
-
Inhibition of Autophagy by Berbamine Hydrochloride Mitigates Tumor Immune Escape by Elevating MHC-I in Melanoma Cells.Cells. 2024 Sep 13;13(18):1537. doi: 10.3390/cells13181537. Cells. 2024. PMID: 39329721 Free PMC article.
-
Enhancement of HLA class II-restricted CD4+ T cell recognition of human melanoma cells following treatment with bryostatin-1.Cell Immunol. 2011;271(2):392-400. doi: 10.1016/j.cellimm.2011.08.007. Epub 2011 Aug 18. Cell Immunol. 2011. PMID: 21903207 Free PMC article.
-
Targeting autophagy as a potential therapeutic approach for melanoma therapy.Semin Cancer Biol. 2013 Oct;23(5):352-60. doi: 10.1016/j.semcancer.2013.06.008. Epub 2013 Jul 2. Semin Cancer Biol. 2013. PMID: 23831275 Review.
Cited by
-
Elevation of c-MYC disrupts HLA class II-mediated immune recognition of human B cell tumors.J Immunol. 2015 Feb 15;194(4):1434-45. doi: 10.4049/jimmunol.1402382. Epub 2015 Jan 16. J Immunol. 2015. PMID: 25595783 Free PMC article.
-
New Potential Pharmacological Functions of Chinese Herbal Medicines via Regulation of Autophagy.Molecules. 2016 Mar 17;21(3):359. doi: 10.3390/molecules21030359. Molecules. 2016. PMID: 26999089 Free PMC article. Review.
-
Autophagy and Programmed Cell Death Modalities Interplay in HIV Pathogenesis.Cells. 2025 Feb 28;14(5):351. doi: 10.3390/cells14050351. Cells. 2025. PMID: 40072080 Free PMC article. Review.
-
Nutritional Profile and Health Benefits of Ganoderma lucidum "Lingzhi, Reishi, or Mannentake" as Functional Foods: Current Scenario and Future Perspectives.Foods. 2022 Apr 1;11(7):1030. doi: 10.3390/foods11071030. Foods. 2022. PMID: 35407117 Free PMC article. Review.
-
Expression Analysis of Autophagy Related Markers LC3B, p62 and HMGB1 Indicate an Autophagy-Independent Negative Prognostic Impact of High p62 Expression in Pulmonary Squamous Cell Carcinomas.Cancers (Basel). 2018 Aug 21;10(9):281. doi: 10.3390/cancers10090281. Cancers (Basel). 2018. PMID: 30134604 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

