Myosin binding protein-C phosphorylation is the principal mediator of protein kinase A effects on thick filament structure in myocardium
- PMID: 22850286
- PMCID: PMC3472100
- DOI: 10.1016/j.yjmcc.2012.07.012
Myosin binding protein-C phosphorylation is the principal mediator of protein kinase A effects on thick filament structure in myocardium
Abstract
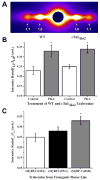
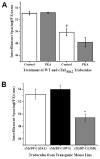
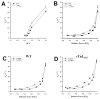
Phosphorylation of cardiac myosin binding protein-C (cMyBP-C) is a regulator of pump function in healthy hearts. However, the mechanisms of regulation by cAMP-dependent protein kinase (PKA)-mediated cMyBP-C phosphorylation have not been completely dissociated from other myofilament substrates for PKA, especially cardiac troponin I (cTnI). We have used synchrotron X-ray diffraction in skinned trabeculae to elucidate the roles of cMyBP-C and cTnI phosphorylation in myocardial inotropy and lusitropy. Myocardium in this study was isolated from four transgenic mouse lines in which the phosphorylation state of either cMyBP-C or cTnI was constitutively altered by site-specific mutagenesis. Analysis of peak intensities in X-ray diffraction patterns from trabeculae showed that cross-bridges are displaced similarly from the thick filament and toward actin (1) when both cMyBP-C and cTnI are phosphorylated, (2) when only cMyBP-C is phosphorylated, and (3) when cMyBP-C phosphorylation is mimicked by replacement with negative charge in its PKA sites. These findings suggest that phosphorylation of cMyBP-C relieves a constraint on cross-bridges, thereby increasing the proximity of myosin to binding sites on actin. Measurements of Ca(2+)-activated force in myocardium defined distinct molecular effects due to phosphorylation of cMyBP-C or co-phosphorylation with cTnI. Echocardiography revealed that mimicking the charge of cMyBP-C phosphorylation protects hearts from hypertrophy and systolic dysfunction that develops with constitutive dephosphorylation or genetic ablation, underscoring the importance of cMyBP-C phosphorylation for proper pump function.
Published by Elsevier Ltd.
Figures
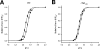
References
-
- Kobayashi T, Solaro RJ. Calcium, thin filaments, and the integrative biology of cardiac contractility. Annu Rev Physiol. 2005;67:39–67. - PubMed
-
- Layland J, Solaro RJ, Shah AM. Regulation of cardiac contractile function by troponin I phosphorylation. Cardiovasc Res. 2005 Apr 1;66(1):12–21. - PubMed
-
- Winegrad S. Cardiac myosin binding protein C. Circ Res. 1999 May 28;84(10):1117–26. - PubMed
-
- Robertson SP, Johnson JD, Holroyde MJ, Kranias EG, Potter JD, Solaro RJ. The effect of troponin I phosphorylation on the Ca2+-binding properties of the Ca2+-regulatory site of bovine cardiac troponin. J Biol Chem. 1982 Jan 10;257(1):260–3. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

