Abiraterone and other novel androgen-directed strategies for the treatment of prostate cancer: a new era of hormonal therapies is born
- PMID: 22852027
- PMCID: PMC3398601
- DOI: 10.1177/1756287212452196
Abiraterone and other novel androgen-directed strategies for the treatment of prostate cancer: a new era of hormonal therapies is born
Abstract
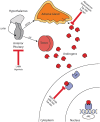
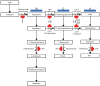
The number of life-prolonging therapies proven effective in the treatment of metastatic castrate-resistant prostate cancer (CRPC) has been limited until recently. In the past 2 years several such therapies have come to market. In 2010, the autologous immunotherapy sipuleucel-T and the next-generation taxane cabazitaxel were approved in this setting. However, abundant evidence has shown that CRPC growth continues to be driven through androgen-dependent signaling. Both of these drugs fail to take advantage of this targetable oncogenic pathway. Potent specific inhibitors of cytochrome P450-17 have been engineered with the aim of suppressing androgen synthesis beyond that seen with the luteinizing hormone-releasing hormone agonists/antagonists. Abiraterone acetate was developed by rational design based on a pregnenolone parent structure. Its approval by the US Food and Drug Administration (FDA) was granted in 2011 based on phase III data demonstrating an overall survival advantage compared with placebo. More recently, other drugs that act along the androgen signaling pathway, such as orteronel (TAK-700), galeterone (TOK-001), enzalutamide (MDV3100) and ARN-509, have shown promise in clinical trials. Some of these are expected to gain FDA approval in the near future. Here, we review abiraterone and other novel androgen-directed therapeutic strategies for the management of advanced prostate cancer.
Keywords: abiraterone; androgen; androgen receptor; enzalutamide; galeterone; orteronel; prostate cancer.
Conflict of interest statement
Figures
References
-
- Agus D., Stadler W., Shevrin L., Hart G., MacVicar O., Hamid J., et al. (2011) Safety, efficacy, and pharmacodynamics of the investigational agent TAK-700 in meta-static castration-resistant prostate cancer (mCRPC): updated data from a phase I/II study. J Clin Oncol 29(Suppl.): abstract 4531
-
- Attard G., Reid A., Auchus R., Hughes B., Cassidy A., Thompson E., et al. (2012) Clinical and biochemical consequences of CYP17A1 inhibition with abiraterone given with and without exogenous glucocorticoids in castrate men with advanced prostate cancer. J Clin Endocrinol Metab 97: 507–516 - PubMed
-
- Attard G., Reid A., Olmos D., De Bono J. (2009a) Antitumor activity with CYP17 blockade indicates that castration-resistant prostate cancer frequently remains hormone driven. Cancer Res 69: 4937–4940 - PubMed
-
- Attard G., Reid A., Yap T., Raynaud F., Dowsett M., Settatree S., et al. (2008) Phase I clinical trial of a selective inhibitor of CYP17, abiraterone acetate, confirms that castration-resistant prostate cancer commonly remains hormone driven. J Clin Oncol 26: 4563–4571 - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

