Novel in situ normal streaming potential device for characterizing electrostatic properties of confluent cells
- PMID: 22852705
- PMCID: PMC6910581
- DOI: 10.1063/1.4737140
Novel in situ normal streaming potential device for characterizing electrostatic properties of confluent cells
Erratum in
- Rev Sci Instrum. 2012 Aug;83(8):089901
Abstract
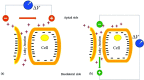
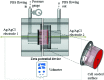
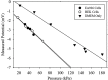
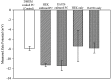
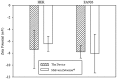
The characteristics of transport across confluent cell monolayers may often be attributed to its electrostatic properties. While tangential streaming potential is often used to quantify these electrostatic properties, this method is not effective for transport normal to the apical cell surface where the charge properties along the basolateral sides may be important (i.e., confluent cells with leaky tight junctions). In addition, even when cells have a uniform charge distribution, the shear stress generated by the conventional tangential flow device may dislodge cells from their confluent state. Here we introduce a novel streaming potential measurement device to characterize the normal electrostatic properties of confluent cells. The streaming potential device encompasses a 24 mm cell-seeded Transwell(®) with two AgCl electrodes on either side of the cell-seeded Transwell. Phosphate buffered saline is pressurized transversal to the Transwell and the resultant pressure gradient induces a potential difference. Confluent monolayers of HEK and EA926 cells are used as examples. The corresponding zeta potential of the cell-membrane configuration is calculated using the Helmholtz-Smoluchowski equation and the zeta potential of the confluent cell layer is deconvolved from the overall measurements. For these test models, the zeta potential is consistent with that determined using a commercial dispersed-cell device. This novel streaming potential device provides a simple, easy, and cost-effective methodology to determine the normal zeta potential of confluent cells cultured on Transwell systems while keeping the cells intact. Furthermore, its versatility allows periodic measurements of properties of the same cell culture during transient studies.
Figures
Similar articles
-
Electrostatic properties of confluent Caco-2 cell layer correlates to their microvilli growth and determines underlying transcellular flow.Biotechnol Bioeng. 2013 Oct;110(10):2742-8. doi: 10.1002/bit.24939. Epub 2013 Jun 27. Biotechnol Bioeng. 2013. PMID: 23613195
-
A chip device to determine surface charge properties of confluent cell monolayers by measuring streaming potential.Lab Chip. 2020 Oct 21;20(20):3792-3805. doi: 10.1039/d0lc00558d. Epub 2020 Sep 11. Lab Chip. 2020. PMID: 32914817
-
Characterizing zeta potential of functional nanofibers in a microfluidic device.J Colloid Interface Sci. 2012 Apr 15;372(1):252-60. doi: 10.1016/j.jcis.2012.01.007. Epub 2012 Jan 20. J Colloid Interface Sci. 2012. PMID: 22305420
-
Status of methods for assessing bacterial cell surface charge properties based on zeta potential measurements.J Microbiol Methods. 2001 Jan;43(3):153-64. doi: 10.1016/s0167-7012(00)00224-4. J Microbiol Methods. 2001. PMID: 11118650 Review.
-
Cell trapping in activated micropores for functional analysis.Conf Proc IEEE Eng Med Biol Soc. 2006;2006:1838-41. doi: 10.1109/IEMBS.2006.260233. Conf Proc IEEE Eng Med Biol Soc. 2006. PMID: 17945673 Review.
Cited by
-
Mapping the electrostatic profiles of cellular membranes.Mol Biol Cell. 2021 Feb 1;32(3):301-310. doi: 10.1091/mbc.E19-08-0436. Epub 2020 Dec 2. Mol Biol Cell. 2021. PMID: 33263429 Free PMC article.
-
Single and binary protein electroultrafiltration using poly(vinyl-alcohol)-carbon nanotube (PVA-CNT) composite membranes.PLoS One. 2020 Apr 16;15(4):e0228973. doi: 10.1371/journal.pone.0228973. eCollection 2020. PLoS One. 2020. PMID: 32298267 Free PMC article.
References
-
- Dekkeq A., Reitsm K., Beugeling T., Bantjeq A., Feijen J., Kirkpatrick C. J., and van Aken W. Gr., Clin. Mater. 11, 157–162 (1992).10.1016/0267-6605(92)90042-R - DOI
-
- Bousse L. J., Mostarshed S., and Hafeman D., Sens. Actuators B 10(1), 67–71 (1992).10.1016/0925-4005(92)80013-N - DOI
-
- Boevé E. R., Cao L. C., De Bruijn W. C., Robertson W. G., Romijn J. C., and Schröder F. H., J. Urol. 152(2), 531–536 (1994). - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

