Cell-adhesive and cell-repulsive zwitterionic oligopeptides for micropatterning and rapid electrochemical detachment of cells
- PMID: 22853640
- PMCID: PMC3530950
- DOI: 10.1089/ten.TEA.2011.0739
Cell-adhesive and cell-repulsive zwitterionic oligopeptides for micropatterning and rapid electrochemical detachment of cells
Abstract
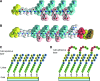
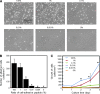
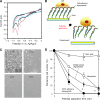
In this study, we describe the development of oligopeptide-modified cell culture surfaces from which adherent cells can be rapidly detached by application of an electrical stimulus. An oligopeptide, CGGGKEKEKEK, was designed with a terminal cysteine residue to mediate binding to a gold surface via a gold-thiolate bond. The peptide forms a self-assembled monolayer through the electrostatic force between the sequence of alternating charged glutamic acid (E) and lysine (K) residues. The dense and electrically neutral oligopeptide zwitterionic layer of the modified surface was resistant to nonspecific adsorption of proteins and adhesion of cells, while the surface was altered to cell adhesive by the addition of a second oligopeptide (CGGGKEKEKEKGRGDSP) containing the RGD cell adhesion motif. Application of a negative electrical potential to this gold surface cleaved the gold-thiolate bond, leading to desorption of the oligopeptide layer, and rapid (within 2 min) detachment of virtually all cells. This approach was applicable not only to detachment of cell sheets but also for transfer of cell micropatterns to a hydrogel. This electrochemical approach of cell detachment may be a useful tool for tissue-engineering applications.
Figures


Similar articles
-
Tissue engineering based on electrochemical desorption of an RGD-containing oligopeptide.J Tissue Eng Regen Med. 2013 Mar;7(3):236-43. doi: 10.1002/term.519. Epub 2011 Dec 12. J Tissue Eng Regen Med. 2013. PMID: 22162306
-
Electrochemical desorption of self-assembled monolayers for engineering cellular tissues.Biomaterials. 2009 Jul;30(21):3573-9. doi: 10.1016/j.biomaterials.2009.03.045. Epub 2009 Apr 10. Biomaterials. 2009. PMID: 19362363
-
Engineering thick cell sheets by electrochemical desorption of oligopeptides on membrane substrates.Regen Ther. 2016 Mar 1;3:24-31. doi: 10.1016/j.reth.2015.12.003. eCollection 2016 Mar. Regen Ther. 2016. PMID: 31245469 Free PMC article.
-
Gold cleaning methods for preparation of cell culture surfaces for self-assembled monolayers of zwitterionic oligopeptides.J Biosci Bioeng. 2018 May;125(5):606-612. doi: 10.1016/j.jbiosc.2017.12.014. Epub 2018 Jan 17. J Biosci Bioeng. 2018. PMID: 29352711
-
The effect of RGD peptide-conjugated magnetite cationic liposomes on cell growth and cell sheet harvesting.Biomaterials. 2005 Nov;26(31):6185-93. doi: 10.1016/j.biomaterials.2005.03.039. Biomaterials. 2005. PMID: 15899515
Cited by
-
Dynamically Tunable Cell Culture Platforms for Tissue Engineering and Mechanobiology.Prog Polym Sci. 2017 Feb;65:53-82. doi: 10.1016/j.progpolymsci.2016.09.004. Epub 2016 Sep 17. Prog Polym Sci. 2017. PMID: 28522885 Free PMC article.
-
Alternating electric field application induced non-contact and enzyme-free cell detachment.Cytotechnology. 2019 Apr;71(2):583-597. doi: 10.1007/s10616-019-00307-4. Epub 2019 Feb 19. Cytotechnology. 2019. PMID: 30783819 Free PMC article.
-
Cell-repellent polyampholyte for conformal coating on microstructures.Sci Rep. 2022 Jun 25;12(1):10815. doi: 10.1038/s41598-022-15177-8. Sci Rep. 2022. PMID: 35752647 Free PMC article.
-
Recapitulating dynamic ECM ligand presentation at biomaterial interfaces: Molecular strategies and biomedical prospects.Exploration (Beijing). 2022 Jan 24;2(1):20210093. doi: 10.1002/EXP.20210093. eCollection 2022 Feb. Exploration (Beijing). 2022. PMID: 37324582 Free PMC article. Review.
-
Tailored cell sheet engineering using microstereolithography and electrochemical cell transfer.Sci Rep. 2019 Jul 18;9(1):10415. doi: 10.1038/s41598-019-46801-9. Sci Rep. 2019. PMID: 31320678 Free PMC article.
References
-
- Langer R. Tirrell D.A. Designing materials for biology and medicine. Nature. 2004;428:487. - PubMed
-
- Guillaume-Gentil O. Gabi M. Zenobi-Wong M. Voros J. Electrochemically switchable platform for the micro-patterning and release of heterotypic cell sheets. Biomed Microdevices. 2011;13:221. - PubMed
-
- Elloumi-Hannachi I. Yamato M. Okano T. Cell sheet engineering: a unique nanotechnology for scaffold-free tissue reconstruction with clinical applications in regenerative medicine. J Intern Med. 2010;267:54. - PubMed
-
- Miyahara Y. Nagaya N. Kataoka M. Yanagawa B. Tanaka K. Hao H., et al. Monolayered mesenchymal stem cells repair scarred myocardium after myocardial infarction. Nat Med. 2006;12:459. - PubMed
-
- Ohashi K. Yokoyama T. Yamato M. Kuge H. Kanehiro H. Tsutsumi M., et al. Engineering functional two- and three-dimensional liver systems in vivo using hepatic tissue sheets. Nat Med. 2007;13:880. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

