Convergent evolution of sodium ion selectivity in metazoan neuronal signaling
- PMID: 22854023
- PMCID: PMC3809514
- DOI: 10.1016/j.celrep.2012.06.016
Convergent evolution of sodium ion selectivity in metazoan neuronal signaling
Abstract
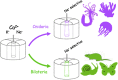
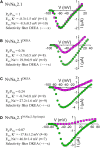
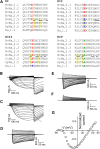
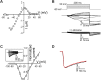
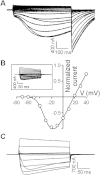
Ion selectivity of metazoan voltage-gated Na(+) channels is critical for neuronal signaling and has long been attributed to a ring of four conserved amino acids that constitute the ion selectivity filter (SF) at the channel pore. Yet, in addition to channels with a preference for Ca(2+) ions, the expression and characterization of Na(+) channel homologs from the sea anemone Nematostella vectensis, a member of the early-branching metazoan phylum Cnidaria, revealed a sodium-selective channel bearing a noncanonical SF. Mutagenesis and physiological assays suggest that pore elements additional to the SF determine the preference for Na(+) in this channel. Phylogenetic analysis assigns the Nematostella Na(+)-selective channel to a channel group unique to Cnidaria, which diverged >540 million years ago from Ca(2+)-conducting Na(+) channel homologs. The identification of Cnidarian Na(+)-selective ion channels distinct from the channels of bilaterian animals indicates that selectivity for Na(+) in neuronal signaling emerged independently in these two animal lineages.
Copyright © 2012 The Authors. Published by Elsevier Inc. All rights reserved.
Figures


Similar articles
-
The evolution of the four subunits of voltage-gated calcium channels: ancient roots, increasing complexity, and multiple losses.Genome Biol Evol. 2014 Aug 21;6(9):2210-7. doi: 10.1093/gbe/evu177. Genome Biol Evol. 2014. PMID: 25146647 Free PMC article.
-
Ether-à-go-go family voltage-gated K+ channels evolved in an ancestral metazoan and functionally diversified in a cnidarian-bilaterian ancestor.J Exp Biol. 2015 Feb 15;218(Pt 4):526-36. doi: 10.1242/jeb.110080. J Exp Biol. 2015. PMID: 25696816 Free PMC article.
-
Evolution of eukaryotic ion channels: principles underlying the conversion of Ca²⁺-selective to Na⁺-selective channels.J Am Chem Soc. 2014 Mar 5;136(9):3553-9. doi: 10.1021/ja4121132. Epub 2014 Feb 20. J Am Chem Soc. 2014. PMID: 24517213
-
Simulation Studies of Ion Permeation and Selectivity in Voltage-Gated Sodium Channels.Curr Top Membr. 2016;78:215-60. doi: 10.1016/bs.ctm.2016.07.005. Epub 2016 Aug 3. Curr Top Membr. 2016. PMID: 27586286 Review.
-
Molecular pore structure of voltage-gated sodium and calcium channels.Braz J Med Biol Res. 1994 Dec;27(12):2781-802. Braz J Med Biol Res. 1994. PMID: 7550000 Review.
Cited by
-
Biophysical characterization of the honeybee DSC1 orthologue reveals a novel voltage-dependent Ca2+ channel subfamily: CaV4.J Gen Physiol. 2016 Aug;148(2):133-45. doi: 10.1085/jgp.201611614. Epub 2016 Jul 18. J Gen Physiol. 2016. PMID: 27432995 Free PMC article.
-
Molecular Characterization of Voltage-Gated Sodium Channels and Their Relations with Paralytic Shellfish Toxin Bioaccumulation in the Pacific Oyster Crassostrea gigas.Mar Drugs. 2017 Jan 19;15(1):21. doi: 10.3390/md15010021. Mar Drugs. 2017. PMID: 28106838 Free PMC article.
-
Normal locomotion in zebrafish lacking the sodium channel NaV1.4 suggests that the need for muscle action potentials is not universal.PLoS Biol. 2025 Apr 24;23(4):e3003137. doi: 10.1371/journal.pbio.3003137. eCollection 2025 Apr. PLoS Biol. 2025. PMID: 40273189 Free PMC article.
-
The evolution of the four subunits of voltage-gated calcium channels: ancient roots, increasing complexity, and multiple losses.Genome Biol Evol. 2014 Aug 21;6(9):2210-7. doi: 10.1093/gbe/evu177. Genome Biol Evol. 2014. PMID: 25146647 Free PMC article.
-
Functional Characterization of Cnidarian HCN Channels Points to an Early Evolution of Ih.PLoS One. 2015 Nov 10;10(11):e0142730. doi: 10.1371/journal.pone.0142730. eCollection 2015. PLoS One. 2015. PMID: 26555239 Free PMC article.
References
-
- Abascal F., Zardoya R., Posada D. ProtTest: selection of best-fit models of protein evolution. Bioinformatics. 2005;21:2104–2105. - PubMed
-
- Anderson P. Properties and pharmacology of a TTX insensitive Na+ current in neurones of the jellyfish Cyanea Capillata. J. Exp. Biol. 1987;133:231–248.
-
- Cai X. Ancient origin of four-domain voltage-gated Na+ channels predates the divergence of animals and fungi. J. Membr. Biol. 2012;245:117–123. - PubMed
Supplemental Reference
-
- Weber, W.-M., Liebold, K.M., Reifarth, F.W., Uhr, U., and Clauss, W. (1995). Influence of extracellular Ca2+ on endogenous Cl- channels in Xenopus oocytes. Pflugers Arch. 429, 820–824. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

