A fast and simple population code for orientation in primate V1
- PMID: 22855811
- PMCID: PMC3506189
- DOI: 10.1523/JNEUROSCI.1335-12.2012
A fast and simple population code for orientation in primate V1
Abstract
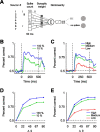
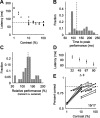
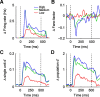
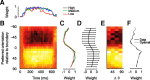
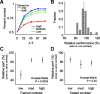
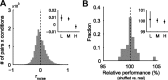
Orientation tuning has been a classic model for understanding single-neuron computation in the neocortex. However, little is known about how orientation can be read out from the activity of neural populations, in particular in alert animals. Our study is a first step toward that goal. We recorded from up to 20 well isolated single neurons in the primary visual cortex of alert macaques simultaneously and applied a simple, neurally plausible decoder to read out the population code. We focus on two questions: First, what are the time course and the timescale at which orientation can be read out from the population response? Second, how complex does the decoding mechanism in a downstream neuron have to be to reliably discriminate between visual stimuli with different orientations? We show that the neural ensembles in primary visual cortex of awake macaques represent orientation in a way that facilitates a fast and simple readout mechanism: With an average latency of 30-80 ms, the population code can be read out instantaneously with a short integration time of only tens of milliseconds, and neither stimulus contrast nor correlations need to be taken into account to compute the optimal synaptic weight pattern. Our study shows that-similar to the case of single-neuron computation-the representation of orientation in the spike patterns of neural populations can serve as an exemplary case for understanding the computations performed by neural ensembles underlying visual processing during behavior.
Figures



Comment in
-
Orienting towards ensembles: from single cells to neural populations.J Neurosci. 2013 Jan 2;33(1):2-3. doi: 10.1523/JNEUROSCI.4658-12.2013. J Neurosci. 2013. PMID: 23283316 Free PMC article. No abstract available.
Similar articles
-
Nature and precision of temporal coding in visual cortex: a metric-space analysis.J Neurophysiol. 1996 Aug;76(2):1310-26. doi: 10.1152/jn.1996.76.2.1310. J Neurophysiol. 1996. PMID: 8871238
-
The linearity and selectivity of neuronal responses in awake visual cortex.J Vis. 2009 Aug 25;9(9):12.1-17. doi: 10.1167/9.9.12. J Vis. 2009. PMID: 19761345 Free PMC article.
-
Coding of stimulus sequences by population responses in visual cortex.Nat Neurosci. 2009 Oct;12(10):1317-24. doi: 10.1038/nn.2398. Epub 2009 Sep 13. Nat Neurosci. 2009. PMID: 19749748 Free PMC article.
-
Fast coding of orientation in primary visual cortex.PLoS Comput Biol. 2012;8(6):e1002536. doi: 10.1371/journal.pcbi.1002536. Epub 2012 Jun 14. PLoS Comput Biol. 2012. PMID: 22719237 Free PMC article.
-
Corticocortical feedback contributes to surround suppression in V1 of the alert primate.J Neurosci. 2013 May 8;33(19):8504-17. doi: 10.1523/JNEUROSCI.5124-12.2013. J Neurosci. 2013. PMID: 23658187 Free PMC article.
Cited by
-
Pattern Motion Direction Is Encoded in the Population Activity of Macaque Area MT.J Neurosci. 2022 Dec 14;42(50):9372-9386. doi: 10.1523/JNEUROSCI.0011-22.2022. Epub 2022 Nov 4. J Neurosci. 2022. PMID: 36332976 Free PMC article.
-
Awareness of the relative quality of spatial working memory representations.Atten Percept Psychophys. 2023 Jul;85(5):1710-1721. doi: 10.3758/s13414-022-02646-5. Epub 2023 Jan 31. Atten Percept Psychophys. 2023. PMID: 36720782 Free PMC article.
-
Linking V1 Activity to Behavior.Annu Rev Vis Sci. 2018 Sep 15;4:287-310. doi: 10.1146/annurev-vision-102016-061324. Epub 2018 Jul 5. Annu Rev Vis Sci. 2018. PMID: 29975592 Free PMC article. Review.
-
The Population Tracking Model: A Simple, Scalable Statistical Model for Neural Population Data.Neural Comput. 2017 Jan;29(1):50-93. doi: 10.1162/NECO_a_00910. Epub 2016 Nov 21. Neural Comput. 2017. PMID: 27870612 Free PMC article.
-
Joint representation of working memory and uncertainty in human cortex.Neuron. 2021 Nov 17;109(22):3699-3712.e6. doi: 10.1016/j.neuron.2021.08.022. Epub 2021 Sep 14. Neuron. 2021. PMID: 34525327 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
