Cross-linking of cell surface amyloid precursor protein leads to increased β-amyloid peptide production in hippocampal neurons: implications for Alzheimer's disease
- PMID: 22855816
- PMCID: PMC3539764
- DOI: 10.1523/JNEUROSCI.6473-11.2012
Cross-linking of cell surface amyloid precursor protein leads to increased β-amyloid peptide production in hippocampal neurons: implications for Alzheimer's disease
Abstract
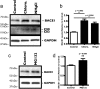
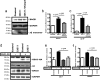
The accumulation of the β-amyloid peptide (Aβ) in Alzheimer's disease (AD) is thought to play a causative role in triggering synaptic dysfunction in neurons, leading to their eventual demise through apoptosis. Aβ is produced and secreted upon sequential cleavage of the amyloid precursor protein (APP) by β-secretases and γ-secretases. However, while Aβ levels have been shown to be increased in the brains of AD patients, little is known about how the cleavage of APP and the subsequent generation of Aβ is influenced, or whether the cleavage process changes over time. It has been proposed that Aβ can bind APP and promote amyloidogenic processing of APP, further enhancing Aβ production. Proof of this idea has remained elusive because a clear mechanism has not been identified, and the promiscuous nature of Aβ binding complicates the task of demonstrating the idea. To work around these problems, we used an antibody-mediated approach to bind and cross-link cell-surface APP in cultured rat primary hippocampal neurons. Here we show that cross-linking of APP is sufficient to raise the levels of Aβ in viable neurons with a concomitant increase in the levels of the β-secretase BACE1. This appears to occur as a result of a sorting defect that stems from the caspase-3-mediated inactivation of a key sorting adaptor protein, namely GGA3, which prevents the lysosomal degradation of BACE1. Together, our data suggest the occurrence of a positive pathogenic feedback loop involving Aβ and APP in affected neurons possibly allowing Aβ to spread to nearby healthy neurons.
Figures







References
-
- Apelt J, Ach K, Schliebs R. Aging-related down-regulation of neprilysin, a putative beta-amyloid-degrading enzyme, in transgenic Tg2576 Alzheimer-like mouse brain is accompanied by an astroglial upregulation in the vicinity of beta-amyloid plaques. Neurosci Lett. 2003;339:183–186. - PubMed
-
- Bertrand E, Brouillet E, Caillé I, Bouillot C, Cole GM, Prochiantz A, Allinquant B. A short cytoplasmic domain of the amyloid precursor protein induces apoptosis in vitro and in vivo. Mol Cell Neurosci. 2001;18:503–511. - PubMed
-
- Blasko I, Beer R, Bigl M, Apelt J, Franz G, Rudzki D, Ransmayr G, Kampfl A, Schliebs R. Experimental traumatic brain injury in rats stimulates the expression, production and activity of Alzheimer's disease beta-secretase (BACE-1) J Neural Transm. 2004;111:523–536. - PubMed
-
- Bonifacino JS. The GGA proteins: adaptors on the move. Nat Rev Mol Cell Biol. 2004;5:23–32. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
