Genetics of gene expression responses to temperature stress in a sea urchin gene network
- PMID: 22856327
- PMCID: PMC3866972
- DOI: 10.1111/j.1365-294X.2012.05717.x
Genetics of gene expression responses to temperature stress in a sea urchin gene network
Abstract
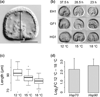
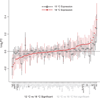
Stress responses play an important role in shaping species distributions and robustness to climate change. We investigated how stress responses alter the contribution of additive genetic variation to gene expression during development of the purple sea urchin, Strongylocentrotus purpuratus, under increased temperatures that model realistic climate change scenarios. We first measured gene expression responses in the embryos by RNA-seq to characterize molecular signatures of mild, chronic temperature stress in an unbiased manner. We found that an increase from 12 to 18 °C caused widespread alterations in gene expression including in genes involved in protein folding, RNA processing and development. To understand the quantitative genetic architecture of this response, we then focused on a well-characterized gene network involved in endomesoderm and ectoderm specification. Using a breeding design with wild-caught individuals, we measured genetic and gene-environment interaction effects on 72 genes within this network. We found genetic or maternal effects in 33 of these genes and that the genetic effects were correlated in the network. Fourteen network genes also responded to higher temperatures, but we found no significant genotype-environment interactions in any of the genes. This absence may be owing to an effective buffering of the temperature perturbations within the network. In support of this hypothesis, perturbations to regulatory genes did not affect the expression of the genes that they regulate. Together, these results provide novel insights into the relationship between environmental change and developmental evolution and suggest that climate change may not expose large amounts of cryptic genetic variation to selection in this species.
© 2012 Blackwell Publishing Ltd.
Figures
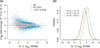



Similar articles
-
The impact of gene expression variation on the robustness and evolvability of a developmental gene regulatory network.PLoS Biol. 2013 Oct;11(10):e1001696. doi: 10.1371/journal.pbio.1001696. Epub 2013 Oct 29. PLoS Biol. 2013. PMID: 24204211 Free PMC article.
-
Genetic variation underlies plastic responses to global change drivers in the purple sea urchin, Strongylocentrotus purpuratus.Proc Biol Sci. 2022 Aug 31;289(1981):20221249. doi: 10.1098/rspb.2022.1249. Epub 2022 Aug 31. Proc Biol Sci. 2022. PMID: 36043281 Free PMC article.
-
Developmental cis-regulatory analysis of the cyclin D gene in the sea urchin Strongylocentrotus purpuratus.Biochem Biophys Res Commun. 2013 Oct 25;440(3):413-8. doi: 10.1016/j.bbrc.2013.09.094. Epub 2013 Oct 1. Biochem Biophys Res Commun. 2013. PMID: 24090975 Free PMC article.
-
Ocean acidification research in the 'post-genomic' era: Roadmaps from the purple sea urchin Strongylocentrotus purpuratus.Comp Biochem Physiol A Mol Integr Physiol. 2015 Jul;185:33-42. doi: 10.1016/j.cbpa.2015.03.007. Epub 2015 Mar 13. Comp Biochem Physiol A Mol Integr Physiol. 2015. PMID: 25773301 Review.
-
Gene regulatory networks for development.Proc Natl Acad Sci U S A. 2005 Apr 5;102(14):4936-42. doi: 10.1073/pnas.0408031102. Epub 2005 Mar 23. Proc Natl Acad Sci U S A. 2005. PMID: 15788537 Free PMC article. Review.
Cited by
-
Without a pinch of salt: effect of low salinity on eggs and nauplii of the salmon louse (Lepeophtheirus salmonis).Parasitol Res. 2023 Aug;122(8):1893-1905. doi: 10.1007/s00436-023-07890-8. Epub 2023 Jun 2. Parasitol Res. 2023. PMID: 37266740 Free PMC article.
-
Robustness and Accuracy in Sea Urchin Developmental Gene Regulatory Networks.Front Genet. 2016 Feb 15;7:16. doi: 10.3389/fgene.2016.00016. eCollection 2016. Front Genet. 2016. PMID: 26913048 Free PMC article.
-
The environmental genomics of metazoan thermal adaptation.Heredity (Edinb). 2015 May;114(5):502-14. doi: 10.1038/hdy.2014.119. Epub 2015 Mar 4. Heredity (Edinb). 2015. PMID: 25735594 Free PMC article. Review.
-
Stressful conditions reveal decrease in size, modification of shape but relatively stable asymmetry in bumblebee wings.Sci Rep. 2018 Oct 11;8(1):15169. doi: 10.1038/s41598-018-33429-4. Sci Rep. 2018. PMID: 30310103 Free PMC article.
-
The Impact of Chronic Heat Stress on the Growth, Survival, Feeding, and Differential Gene Expression in the Sea Urchin Strongylocentrotus intermedius.Front Genet. 2019 Apr 4;10:301. doi: 10.3389/fgene.2019.00301. eCollection 2019. Front Genet. 2019. PMID: 31019527 Free PMC article.
References
-
- Abramoff MD, Magelhaes PJ, Ram SJ. Image processing with ImageJ. Biophotonics international. 2004;11:36–42.
-
- Andronikov V. Heat resistance of gametes of marine invertebrates in relation to temperature conditions under which the species exist. Marine Biology. 1975;30:1–11.
-
- Angerer LM, Angerer RC. Patterning the Sea urchin embryo: gene regulatory networks, signaling pathways, and cellular interactions. Current Topics in Developmental Biology. 2003;53:159–198. - PubMed
-
- Azad AK, Pearce CM, McKinley RS. Influence of stocking density and temperature on early development and survival of the purple sea urchin, Strongylocentrotus purpuratus (Stimpson, 1857) Aquaculture Research. 2011:1–15.
-
- Barry JP, Baxter CH, Sagarin RD, Gilman SE. Climate-related, long-term faunal changes in a California rocky intertidal community. Science. 1995;267:672–675. - PubMed
Publication types
MeSH terms
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

