Autophagy in pancreatic cancer pathogenesis and treatment
- PMID: 22860230
- PMCID: PMC3410583
Autophagy in pancreatic cancer pathogenesis and treatment
Abstract
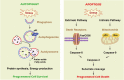
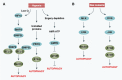
Pancreatic cancer is the fourth most common cancer to cause death due to advanced stage at diagnosis and poor response to current treatment. Autophagy is the lysosome-mediated degradation pathway which plays a critical role in cellular defense, quality control, and energy metabolism. Targeting autophagy is now an exciting field for translational cancer research, as autophagy dysfunction is among the hallmarks of cancer. Pancreatic tumors have elevated autophagy under basal conditions when compared with other epithelial cancers. This review describes our current understanding of the interaction between autophagy and pancreatic cancer development, including risk factors (e.g., pancreatitis, smoking, and alcohol use), tumor microenvironment (e.g., hypoxia and stromal cells), and molecular biology (e.g., K-Ras and p53) of pancreatic cancer. The importance of the HMGB1-RAGE pathway in regulation of autophagy and pancreatic cancer is also presented. Finally, we describe current studies involving autophagy inhibition using either pharmacological inhibitors (e.g., chloroquine) or RNA interference of essential autophagy genes that regulate chemotherapy sensitivity in pancreatic cancer. Summarily, autophagy plays multiple roles in the regulation of pancreatic cancer pathogenesis and treatment, although the exact mechanisms remain unknown.
Keywords: AMPK; Autophagy; HIF1α; HMGB1; RAGE; hypoxia; oncogene; p53; pancreatic cancer; pancreatitis.
Figures


Similar articles
-
Autophagy in cancer associated fibroblasts promotes tumor cell survival: Role of hypoxia, HIF1 induction and NFκB activation in the tumor stromal microenvironment.Cell Cycle. 2010 Sep 1;9(17):3515-33. doi: 10.4161/cc.9.17.12928. Epub 2010 Sep 9. Cell Cycle. 2010. PMID: 20855962 Free PMC article.
-
UBL4A inhibits autophagy-mediated proliferation and metastasis of pancreatic ductal adenocarcinoma via targeting LAMP1.J Exp Clin Cancer Res. 2019 Jul 9;38(1):297. doi: 10.1186/s13046-019-1278-9. J Exp Clin Cancer Res. 2019. PMID: 31288830 Free PMC article.
-
The marine natural product Scalarin inhibits the receptor for advanced glycation end products (RAGE) and autophagy in the PANC-1 and MIA PaCa-2 pancreatic cancer cell lines.Invest New Drugs. 2019 Apr;37(2):262-270. doi: 10.1007/s10637-018-0635-4. Epub 2018 Jul 12. Invest New Drugs. 2019. PMID: 29998364 Free PMC article.
-
Epidemiological-molecular evidence of metabolic reprogramming on proliferation, autophagy and cell signaling in pancreas cancer.Cancer Lett. 2015 Jan 28;356(2 Pt A):281-8. doi: 10.1016/j.canlet.2014.03.028. Epub 2014 Apr 2. Cancer Lett. 2015. PMID: 24704294 Review.
-
[Autophagy contributes to the initiation of pancreatic cancer].Med Sci (Paris). 2017 Mar;33(3):335-339. doi: 10.1051/medsci/20173303022. Epub 2017 Apr 3. Med Sci (Paris). 2017. PMID: 28367822 Review. French.
Cited by
-
Encouraging long-term survival following autophagy inhibition using neoadjuvant hydroxychloroquine and gemcitabine for high-risk patients with resectable pancreatic carcinoma.Cancer Med. 2021 Oct;10(20):7233-7241. doi: 10.1002/cam4.4211. Epub 2021 Sep 24. Cancer Med. 2021. PMID: 34559451 Free PMC article. Review.
-
Cymantrenyl-Nucleobases: Synthesis, Anticancer, Antitrypanosomal and Antimicrobial Activity Studies.Molecules. 2017 Dec 14;22(12):2220. doi: 10.3390/molecules22122220. Molecules. 2017. PMID: 29240697 Free PMC article.
-
Lysosomal dysfunction and autophagy blockade contribute to IMB-6G-induced apoptosis in pancreatic cancer cells.Sci Rep. 2017 Jan 31;7:41862. doi: 10.1038/srep41862. Sci Rep. 2017. PMID: 28139733 Free PMC article.
-
Tumors Responsive to Autophagy-Inhibition: Identification and Biomarkers.Cancers (Basel). 2020 Aug 31;12(9):2463. doi: 10.3390/cancers12092463. Cancers (Basel). 2020. PMID: 32878084 Free PMC article. Review.
-
RAGE Up-Regulation Differently Affects Cell Proliferation and Migration in Pancreatic Cancer Cells.Int J Mol Sci. 2020 Oct 19;21(20):7723. doi: 10.3390/ijms21207723. Int J Mol Sci. 2020. PMID: 33086527 Free PMC article.
References
-
- Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011: The impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin. 2011;61:212–236. - PubMed
-
- Wang Z, Li Y, Ahmad A, Banerjee S, Azmi AS, Kong D, Sarkar FH. Pancreatic cancer: understanding and overcoming chemoresistance. Nat Rev Gastroenterol Hepatol. 2011;8:27–33. - PubMed
-
- Hidalgo M. Pancreatic cancer. N Engl J Med. 2010;362:1605–1617. - PubMed
-
- Hruban RH, Adsay NV. Molecular classification of neoplasms of the pancreas. Hum Pathol. 2009;40:612–623. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
