Pathogenetic and predictive value of biomarkers in patients with ALI and lower severity of illness: results from two clinical trials
- PMID: 22865551
- PMCID: PMC3469636
- DOI: 10.1152/ajplung.00195.2012
Pathogenetic and predictive value of biomarkers in patients with ALI and lower severity of illness: results from two clinical trials
Abstract
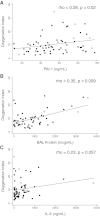
Plasma and bronchoalveolar lavage (BAL) biomarkers related to the pathogenesis of acute lung injury (ALI) have previously been associated with poorer clinical outcomes and increased disease severity among patients with ALI. Whether these biomarkers have predictive value in a less severely ill population that excludes septic patients with high APACHE II scores is currently unknown. We tested the association of plasma and BAL biomarkers with physiological markers of ALI severity or clinically relevant outcomes in a secondary analysis of a clinical trial of activated protein C for the treatment of ALI. Plasma plasminogen activator inhibitor-1 (PAI-1) and mini-BAL protein were both significantly associated with increased oxygenation index (P = 0.02 and 0.01, respectively), whereas there was a trend toward an association between IL-6 and oxygenation index (P = 0.057). High plasma IL-6, thrombomodulin, and mini-BAL protein were all significantly associated with fewer ventilator-free days (VFDs) (P = 0.01, 0.01, and 0.05, respectively); no markers were associated with mortality, but we hypothesized that this was due to the small size of our cohort and the low death rate. To confirm these associations in a larger sample, we identified a restricted cohort of patients from the ARDS Network ALVEOLI study with similar baseline characteristics. We retested the associations of the significant biomarkers with markers of severity and clinical outcomes and studied IL-8 as an additional biomarker given its important predictive value in prior studies. In this restricted cohort, IL-6 was significantly associated with oxygenation index (P = 0.02). Both IL-6 and IL-8 were associated with decreased VFDs and increased 28-day mortality. Future studies should be focused on examining larger numbers of patients with less severe ALI to further test the relative predictive value of plasma and mini-BAL biomarkers for clinically relevant outcomes, including VFDs and mortality, and for their prospective utility in risk stratification for future clinical trials.
Figures
References
-
- Acute Respiratory Distress Syndrome Network Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury, and the acute respiratory distress syndrome. N Engl J Med 342: 1301–1308, 2000 - PubMed
-
- Bernard GR, Artigas A, Brigham KL, Carlet J, Falke K, Hudson L, Lamy M, Legall JR, Morris A, Spragg R. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 149: 818–824, 1994 - PubMed
-
- Brower R, Lanken P, MacIntyre N, Matthay M, Morris A, Ancukiewicz M, Schoenfeld D, Thompson B; National Heart, Lung, and Blood Institute ARDS Clinical Trials Network Higher versus lower positive end-expiratory pressures in patients with the acute respiratory distress syndrome. N Engl J Med 351: 327–336, 2004 - PubMed
-
- Calfee CS, Budev MM, Matthay MA, Church G, Brady S, Uchida T, Ishizaka A, Lara A, Ranes JL, deCamp MM, Arroliga AC. Plasma receptor for advanced glycation end-products predicts duration of ICU stay and mechanical ventilation in patients after lung transplantation. J Heart Lung Transplant 26: 675–680, 2007 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- N01-HR 46063/HR/NHLBI NIH HHS/United States
- HL51856/HL/NHLBI NIH HHS/United States
- 8 K12 RR023262/RR/NCRR NIH HHS/United States
- N01-HR 46060/HR/NHLBI NIH HHS/United States
- N01-HR 46057/HR/NHLBI NIH HHS/United States
- N01-HR 46061/HR/NHLBI NIH HHS/United States
- N01-HR 46054/HR/NHLBI NIH HHS/United States
- N01-HR 46059/HR/NHLBI NIH HHS/United States
- K12 RR023262/RR/NCRR NIH HHS/United States
- N01-HR 46062/HR/NHLBI NIH HHS/United States
- N01-HR 46055/HR/NHLBI NIH HHS/United States
- TL1 RR024129/RR/NCRR NIH HHS/United States
- N01-HR 46056/HR/NHLBI NIH HHS/United States
- N01-HR 46058/HR/NHLBI NIH HHS/United States
- HL74005/HL/NHLBI NIH HHS/United States
- N01-HR 46064/HR/NHLBI NIH HHS/United States
- R37 HL051856/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

