Light-triggered disassembly of amyloid fibrils
- PMID: 22867440
- PMCID: PMC3432263
- DOI: 10.1021/la302626d
Light-triggered disassembly of amyloid fibrils
Abstract
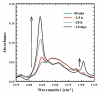
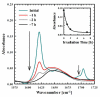
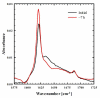
There is growing demand for novel methods that could render the controlled disassembly of higher-order structures formed, for example, by peptides. Herein, we demonstrate such a method based on the application of a photocaged variant of the amino acid lysine, namely, lys(Nvoc). Specifically, we introduce lys(Nvoc) into the primary sequence of the amyloidogenic peptide, Aβ(16-22), at a position where the native side chain is known to play a key role in fibril formation via hydrophobic interactions. Both AFM and infrared spectroscopic measurements indicate that the resultant Aβ(16-22) mutant is able to form fibrils whereas, more importantly, the fibrils thus formed can be completely disassembled upon irradiation with near-UV light, which cleaves the photolabile Nvoc moiety and triggers the restoration of the lysine side chain. These results suggest that the generation of a single charge in a highly hydrophobic region of the fibrils is sufficient to promote their dissociation. Thus, we envisage that the current approach will find useful applications wherein controlled structural disassembly or content release is required.
Figures



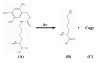
Similar articles
-
N-terminal lipid conjugation of amyloid β(1-40) leads to the formation of highly ordered N-terminally extended fibrils.Phys Chem Chem Phys. 2017 Jan 18;19(3):1839-1846. doi: 10.1039/c6cp05982a. Phys Chem Chem Phys. 2017. PMID: 28000812
-
Effect of introducing a short amyloidogenic sequence from the Aβ peptide at the N-terminus of 18-residue amphipathic helical peptides.J Pept Sci. 2012 Feb;18(2):122-8. doi: 10.1002/psc.1424. Epub 2011 Nov 3. J Pept Sci. 2012. PMID: 22052825
-
Structure-activity relationships in peptide modulators of β-amyloid protein aggregation: variation in α,α-disubstitution results in altered aggregate size and morphology.ACS Chem Neurosci. 2010 Sep 15;1(9):608-26. doi: 10.1021/cn100045q. Epub 2010 Jul 8. ACS Chem Neurosci. 2010. PMID: 22778850 Free PMC article.
-
Structural origin of polymorphism of Alzheimer's amyloid β-fibrils.Biochem J. 2012 Oct 1;447(1):43-50. doi: 10.1042/BJ20120034. Biochem J. 2012. PMID: 22823461
-
New Mechanism of Amyloid Fibril Formation.Curr Protein Pept Sci. 2019;20(6):630-640. doi: 10.2174/1389203720666190125160937. Curr Protein Pept Sci. 2019. PMID: 30686252 Review.
Cited by
-
Ultrasound-facilitated assembly and disassembly of a pH-sensitive self-assembly peptide.RSC Adv. 2018 Aug 20;8(51):29482-29487. doi: 10.1039/c8ra04391d. eCollection 2018 Aug 14. RSC Adv. 2018. PMID: 35548023 Free PMC article.
-
Specificity from nonspecific interaction: regulation of tumor necrosis factor-α activity by DNA.J Biol Chem. 2019 Apr 19;294(16):6397-6404. doi: 10.1074/jbc.RA119.007586. Epub 2019 Feb 27. J Biol Chem. 2019. PMID: 30814250 Free PMC article.
-
Possible Existence of α-Sheets in the Amyloid Fibrils Formed by a TTR105-115 Mutant.J Am Chem Soc. 2018 Jan 17;140(2):629-635. doi: 10.1021/jacs.7b09262. Epub 2018 Jan 4. J Am Chem Soc. 2018. PMID: 29241000 Free PMC article.
-
Light-induced modulation of DNA recognition by the Rad4/XPC damage sensor protein.RSC Chem Biol. 2021 Apr 1;2(2):523-536. doi: 10.1039/d0cb00192a. Epub 2021 Jan 6. RSC Chem Biol. 2021. PMID: 34041491 Free PMC article.
-
Amide I Band and Photoinduced Disassembly of a Peptide Hydrogel.Chem Phys Lett. 2013 Aug 6;580:135-140. doi: 10.1016/j.cplett.2013.06.055. Chem Phys Lett. 2013. PMID: 23997272 Free PMC article.
References
-
- Chiti F, Dobson CM. Protein Misfolding, Functional Amyloid, and Human Disease. Ann. Rev. Biochem. 2006;75:333–366. - PubMed
-
- Cherny I;, Gazit E. Amyloid: Not Only Pathological Agents but Also Ordered Nanomaterials. Angew. Chem. Int. Ed. 2008;47:4062–4069. - PubMed
-
- Knowles TPJ, Buehler MJ. Nanomechanics of Functional and Pathological Amyloid Materials. Nature Nanotech. 2011;6:469–479. - PubMed
-
- Waterhouse SH, Gerrard JA. Amyloid Fibrils in Biotechnology. Aust. J. Chem. 2004;57:519–523.
-
- Rajagopal K, Schneider J. Self-Assembling Peptides and Proteins for Nanotechnological Applications. Curr. Opin. Struct. Biol. 2004;14:480–486. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

