Herb-drug interaction between Echinacea purpurea and etravirine in HIV-infected patients
- PMID: 22869560
- PMCID: PMC3457385
- DOI: 10.1128/AAC.01205-12
Herb-drug interaction between Echinacea purpurea and etravirine in HIV-infected patients
Abstract
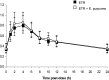
The aim of this open-label, fixed-sequence study was to investigate the potential of the botanical supplement Echinacea purpurea to interact with etravirine, a nonnucleoside reverse transcriptase inhibitor of HIV. Fifteen HIV-infected patients receiving antiretroviral therapy with etravirine (400 mg once daily) for at least 4 weeks were included. E. purpurea root/extract-containing capsules were added to the antiretroviral treatment (500 mg every 8 h) for 14 days. Etravirine concentrations in plasma were determined by high-performance liquid chromatography immediately before and 1, 2, 4, 6, 8, 10, 12, and 24 h after a morning dose of etravirine on day 0 and etravirine plus E. purpurea on day 14. Individual etravirine pharmacokinetic parameters were calculated by noncompartmental analysis and compared between days 0 and 14 by means of the geometric mean ratio (GMR) and its 90% confidence interval (CI). The median age was 46 years (interquartile range, 41 to 50), and the median body weight was 76 kg (interquartile range, 68 to 92). Echinacea was well tolerated, and all participants completed the study. The GMR for etravirine coadministered with E. purpurea relative to etravirine alone was 1.07 (90% CI, 0.81 to 1.42) for the maximum concentration, 1.04 (90% CI, 0.79 to 1.38) for the area under the concentration-time curve from 0 to 24 h, and 1.04 (90% CI, 0.74 to 1.44) for the concentration at the end of the dosing interval. In conclusion, the coadministration of E. purpurea with etravirine was safe and well tolerated in HIV-infected patients; our data suggest that no dose adjustment for etravirine is necessary.
Figures

References
-
- Barnes J, Anderson LA, Gibbons S, Phillipson JD. 2005. Echinacea species [Echinacea angustifolia (DC.) Hell., Echinacea pallida (Nutt.) Nutt., Echinacea purpurea (L.) Moench]: a review of their chemistry, pharmacology and clinical properties. J. Pharm. Pharmacol. 57:929–954 - PubMed
-
- Barnes PM, Powell-Griner E, McFann K, Nahin RL. 2004. Complementary and alternative medicine use among adults: United States, 2002, p 1–19 Adv. Data, no. 343 - PubMed
-
- Blumenthal M. 1999. Herb market levels after five years of boom, p 64–65 HerbalGram, no. 47
-
- Boffito M, et al. 2009. Pharmacokinetics and safety of etravirine administered once or twice daily after 2 weeks treatment with efavirenz in healthy volunteers. J. Acquir. Immune Defic. Syndr. 52:222–227 - PubMed
-
- DeJesus E, et al. 2010. Pharmacokinetics of once-daily etravirine without and with once-daily darunavir/ritonavir in antiretroviral-naive HIV type-1-infected adults. Antivir. Ther. 15:711–720 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

