A role for NT-3 in the hyperinnervation of neonatally wounded skin
- PMID: 22871470
- PMCID: PMC3657181
- DOI: 10.1016/j.pain.2012.07.012
A role for NT-3 in the hyperinnervation of neonatally wounded skin
Abstract
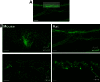
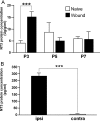
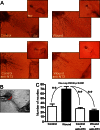
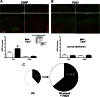
Neurotrophin-3 (NT-3) is a target-derived neurotrophic factor that regulates sensory neuronal survival and growth. Here we report that NT-3 plays a critical permissive role in cutaneous sensory nerve sprouting that contributes to pain and sensitivity following skin wounding in young animals. Sensory terminal sprouting in neonatally wounded dermis and epidermis is accompanied by increased NT-3 transcription, NT-3 protein levels, and NT-3 protein release 3-7 days post skin injury in newborn rats and mice. Functional blockade of NT-3 activity with specific antibodies greatly reduces sensory neurite outgrowth induced by wounded skin, but not by naïve skin, in dorsal root ganglion/skin co-cultures. The requirement for NT-3 for sensory terminal sprouting in vivo is confirmed by the absence of wound-induced hyperinnervation in heterozygous transgenic mice (NT-3(+/-)lacZ). We conclude that upregulation of NT-3 in neonatally wounded skin is a critical factor mediating the sensory nerve sprouting that underlies hypersensitivity and pain following skin injury.
Copyright © 2012 International Association for the Study of Pain. Published by Elsevier B.V. All rights reserved.
Figures


References
-
- Airaksinen M.S., Koltzenburg M., Lewin G.R., Masu Y., Helbig C., Wolf E., Brem G., Toyka K.V., Thoenen H., Meyer M. Specific subtypes of cutaneous mechanoreceptors require neurotrophin-3 following peripheral target innervation. Neuron. 1996;16:287–295. - PubMed
-
- Albers K.M., Davis B.M. The skin as a neurotrophic organ. Neuroscientist. 2007;13:371–382. - PubMed
-
- Alvares D., Torsney C., Beland B., Reynolds M., Fitzgerald M. Modelling the prolonged effects of neonatal pain. Prog Brain Res. 2000;129:365–373. - PubMed
-
- Anand K.J. Pain, plasticity, and premature birth: a prescription for permanent suffering? Nat Med. 2000;6:971–973. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

