Presynaptic GABA(B) receptors decrease neurotransmitter release in vestibular nuclei neurons during vestibular compensation
- PMID: 22871524
- PMCID: PMC4106701
- DOI: 10.1016/j.neuroscience.2012.07.061
Presynaptic GABA(B) receptors decrease neurotransmitter release in vestibular nuclei neurons during vestibular compensation
Abstract
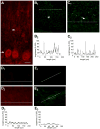
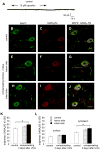
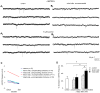
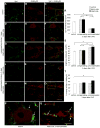
Unilateral damage to the peripheral vestibular receptors precipitates a debilitating syndrome of oculomotor and balance deficits at rest, which extensively normalize during the first week after the lesion due to vestibular compensation. In vivo studies suggest that GABA(B) receptor activation facilitates recovery. However, the presynaptic or postsynaptic sites of action of GABA(B) receptors in vestibular nuclei neurons after lesions have not been determined. Accordingly, here presynaptic and postsynaptic GABA(B) receptor activity in principal cells of the tangential nucleus, a major avian vestibular nucleus, was investigated using patch-clamp recordings correlated with immunolabeling and confocal imaging of the GABA(B) receptor subunit-2 (GABA(B)R2) in controls and operated chickens shortly after unilateral vestibular ganglionectomy (UVG). Baclofen, a GABA(B) agonist, generated no postsynaptic currents in principal cells in controls, which correlated with weak GABA(B)R2 immunolabeling on principal cell surfaces. However, baclofen decreased miniature excitatory postsynaptic current (mEPSC) and GABAergic miniature inhibitory postsynaptic current (mIPSC) events in principal cells in controls, compensating and uncompensated chickens three days after UVG, indicating the presence of functional GABA(B) receptors on presynaptic terminals. Baclofen decreased GABAergic mIPSC frequency to the greatest extent in principal cells on the intact side of compensating chickens, with concurrent increases in GABA(B)R2 pixel brightness and percentage overlap in synaptotagmin 2-labeled terminals. In uncompensated chickens, baclofen decreased mEPSC frequency to the greatest extent in principal cells on the intact side, with concurrent increases in GABA(B)R2 pixel brightness and percentage overlap in synaptotagmin 1-labeled terminals. Altogether, these results revealed changes in presynaptic GABA(B) receptor function and expression which differed in compensating and uncompensated chickens shortly after UVG. This work supports an important role for GABA(B) autoreceptor-mediated inhibition in vestibular nuclei neurons on the intact side during early stages of vestibular compensation, and a role for GABA(B) heteroreceptor-mediated inhibition of glutamatergic terminals on the intact side in the failure to recover function.
Copyright © 2012 IBRO. Published by Elsevier Ltd. All rights reserved.
Figures


Similar articles
-
Presynaptic and postsynaptic ion channel expression in vestibular nuclei neurons after unilateral vestibular deafferentation.J Vestib Res. 2009;19(5-6):191-200. doi: 10.3233/VES-2009-0348. J Vestib Res. 2009. PMID: 20495236 Review.
-
Adaptation of chicken vestibular nucleus neurons to unilateral vestibular ganglionectomy.Neuroscience. 2009 Jul 21;161(4):988-1007. doi: 10.1016/j.neuroscience.2009.04.027. Epub 2009 Apr 15. Neuroscience. 2009. PMID: 19375485
-
GABAB receptors in the medial septum/diagonal band slice from 16-25 day rat.Neuroscience. 2005;132(3):789-800. doi: 10.1016/j.neuroscience.2005.01.027. Neuroscience. 2005. PMID: 15837139
-
Plasticity of spontaneous excitatory and inhibitory synaptic activity in morphologically defined vestibular nuclei neurons during early vestibular compensation.J Neurophysiol. 2012 Jan;107(1):29-41. doi: 10.1152/jn.00406.2011. Epub 2011 Sep 28. J Neurophysiol. 2012. PMID: 21957228 Free PMC article.
-
Key differences in regulation of opioid receptors localized to presynaptic terminals compared to somas: Relevance for novel therapeutics.Neuropharmacology. 2023 Mar 15;226:109408. doi: 10.1016/j.neuropharm.2022.109408. Epub 2022 Dec 28. Neuropharmacology. 2023. PMID: 36584882 Free PMC article. Review.
Cited by
-
A New Model for Congenital Vestibular Disorders.J Assoc Res Otolaryngol. 2019 Apr;20(2):133-149. doi: 10.1007/s10162-018-00705-z. Epub 2018 Dec 18. J Assoc Res Otolaryngol. 2019. PMID: 30564989 Free PMC article.
-
Plastic Events of the Vestibular Nucleus: the Initiation of Central Vestibular Compensation.Mol Neurobiol. 2024 Nov;61(11):9680-9693. doi: 10.1007/s12035-024-04208-2. Epub 2024 Apr 30. Mol Neurobiol. 2024. PMID: 38689145 Review.
-
Expression and Neurotransmitter Association of the Synaptic Calcium Sensor Synaptotagmin in the Avian Auditory Brain Stem.J Assoc Res Otolaryngol. 2022 Dec;23(6):701-720. doi: 10.1007/s10162-022-00863-1. Epub 2022 Aug 23. J Assoc Res Otolaryngol. 2022. PMID: 35999323 Free PMC article.
-
Understanding the Pathophysiology of Congenital Vestibular Disorders: Current Challenges and Future Directions.Front Neurol. 2021 Sep 10;12:708395. doi: 10.3389/fneur.2021.708395. eCollection 2021. Front Neurol. 2021. PMID: 34589045 Free PMC article. Review.
-
Drug-Drug Interactions in Vestibular Diseases, Clinical Problems, and Medico-Legal Implications.Int J Environ Res Public Health. 2021 Dec 8;18(24):12936. doi: 10.3390/ijerph182412936. Int J Environ Res Public Health. 2021. PMID: 34948545 Free PMC article. Review.
References
-
- Akbarian S, Grusser OJ, Guldin WO. Corticofugal projections to the vestibular nucleuin squirrel monkeys: further evidence of multiple cortical vestibular fields. J Comp Neurol. 1993;332:89–104. - PubMed
-
- Akhtar ND, Land PW. Activity-dependent regulation of glutamic acid decarboxylase in the rat barrel cortex: effects of neonatal versus adult sensory deprivation. J Comp Neurol. 1991;307:200–213. - PubMed
-
- Aldrich EM, Peusner KD. Vestibular compensation after ganglionectomy: ultrastructural study of the tangential vestibular nucleus and behavioral study of the hatchling chick. J Neurosci Res. 2002;67:122–138. - PubMed
-
- Astori S, Luján R, K hr G. GABA release from cerebellar stellate cells is developmentally regulated by presynaptic GABA (B) receptors in a target-cell-specific manner. Eur J Neurosci. 2009;30:551–559. - PubMed
-
- Badke MB, Pyle GM, Shea T, Miedaner J. Outcomes in vestibular ablative procedures. Otol Neurotol. 2002;23:504–509. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

