Systemic corticosteroid monotherapy for clinically diagnosed acute rhinosinusitis: a randomized controlled trial
- PMID: 22872770
- PMCID: PMC3470641
- DOI: 10.1503/cmaj.120430
Systemic corticosteroid monotherapy for clinically diagnosed acute rhinosinusitis: a randomized controlled trial
Abstract
Background: Patients with acute rhinosinusitis are frequently encountered in primary care. Although corticosteroids are being increasingly used for symptom control, evidence supporting their use is inconclusive. We conducted a randomized controlled trial to examine the effectiveness of systemic corticosteroid monotherapy for clinically diagnosed, uncomplicated acute rhinosinusitis.
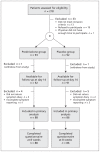
Methods: We conducted a block-randomized, double-blind, placebo-controlled clinical trial at 54 primary care practices (68 family physicians) in the Netherlands between Dec. 30, 2008, and Apr. 28, 2011. Adult patients with clinically diagnosed acute rhinosinusitis were randomly assigned to receive either prednisolone 30 mg/d or placebo for 7 days and asked to complete a symptom diary for 14 days. The primary outcome measure was the proportion of patients with resolution of facial pain or pressure on day 7.
Results: Of the 185 patients included in the trial (93 in the treatment group, 92 in the placebo group), 2 withdrew from the study and 9 were excluded from the primary analysis because of incomplete symptom reporting. The remaining 174 patients (88 in the treatment group, 86 in the placebo group) were included in the intention-to-treat analysis. The proportions of patients with resolution of facial pain or pressure on day 7 were 62.5% (55/88) in the prednisolone group and 55.8% (48/86) in the placebo group (absolute risk difference 6.7%, 95% confidence interval -7.9% to 21.2%). The groups were similar with regard to the decrease over time in the proportion of patients with total symptoms (combined symptoms of runny nose, postnasal discharge, nasal congestion, cough and facial pain) and health-related quality of life. Adverse events were mild and did not differ significantly between the groups.
Interpretation: Systemic corticosteroid monotherapy had no clinically relevant beneficial effects among patients with clinically diagnosed acute rhinosinusitis. Netherlands Trial Register registration no. 1295 (www.trialregister.nl/trialreg/index.asp).
Figures
Comment in
-
Acute rhinosinusitis and systemic corticosteroids.CMAJ. 2013 Jan 8;185(1):62. doi: 10.1503/cmaj.113-2092. CMAJ. 2013. PMID: 23297139 Free PMC article. No abstract available.
-
Acute rhinosinusitis and systemic corticosteroids--response to Ukwaja.CMAJ. 2013 Jan 8;185(1):62. doi: 10.1503/cmaj.113-2093. CMAJ. 2013. PMID: 23297140 Free PMC article. No abstract available.
References
-
- Kuyvenhoven M, van Essen G, Schellevis F, et al. Management of upper respiratory tract infections in Dutch general practice; antibiotic prescribing rates and incidences in 1987 and 2001. Fam Pract 2006;23:175–9 - PubMed
-
- Fokkens W, Lund V, Mullol J. European position paper on rhinosinusitis and nasal polyps 2007. Rhinol Suppl 2007:1–136 - PubMed
-
- Laine K, Maatta T, Varonen H, et al. Diagnosing acute maxillary sinusitis in primary care: a comparison of ultrasound, clinical examination and radiography. Rhinology 1998;36:2–6 - PubMed
-
- Lindbaek M, Hjortdahl P, Johnsen UL. Use of symptoms, signs, and blood tests to diagnose acute sinus infections in primary care: comparison with computed tomography. Fam Med 1996;28:183–8 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous