Minor activities and transition state properties of the human steroid hydroxylases cytochromes P450c17 and P450c21, from reactions observed with deuterium-labeled substrates
- PMID: 22873692
- PMCID: PMC3471366
- DOI: 10.1021/bi300895w
Minor activities and transition state properties of the human steroid hydroxylases cytochromes P450c17 and P450c21, from reactions observed with deuterium-labeled substrates
Abstract
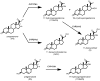
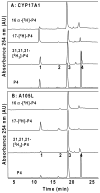
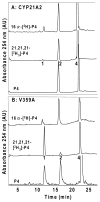
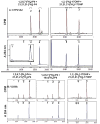
The steroid hydroxylases CYP17A1 (P450c17, 17-hydroxylase/17,20-lyase) and CYP21A2 (P450c21, 21-hydroxylase) catalyze progesterone hydroxylation at one or more sites within a 2 Å radius. We probed their hydrogen atom abstraction mechanisms and regiochemical plasticity with deuterium-labeled substrates: 17-[(2)H]-pregnenolone; 17-[(2)H]-, 16α-[(2)H]-, 21,21,21-[(2)H(3)]-, and 21-[(2)H]-progesterone; and 21,21,21-[(2)H(3)]-17-hydroxyprogesterone. Product distribution and formation rates with recombinant human P450-oxidoreductase and wild-type human CYP17A1 or mutation A105L (reduced progesterone 16α-hydroxylation) and wild-type human CYP21A2 or mutation V359A (substantial progesterone 16α-hydroxylation) were used to calculate intramolecular and intermolecular kinetic isotope effects (KIEs). The intramolecular KIEs for CYP17A1 and mutation A105L were 4.1 and 3.8, respectively, at H-17 and 2.9 and 5.1, respectively, at H-16α. Mutation A105L 21-hydroxylates progesterone (5% of products), and wild-type CYP17A1 also catalyzes a trace of 21-hydroxylation, which increases with 16α-[(2)H]- and 17-[(2)H]-progesterone. The intramolecular KIEs with CYP21A2 mutation V359A and progesterone were 6.2 and 3.8 at H-21 and H-16α, respectively. Wild-type CYP21A2 also forms a trace of 16α-hydroxyprogesterone, which increased with 21,21,21-[(2)H(3)]-progesterone substrate. Competitive intermolecular KIEs paralleled the intramolecular KIE values, with (D)V values of 1.4-5.1 and (D)V/K values of 1.8-5.1 for these reactions. CYP17A1 and CYP21A2 mutation V359A both 16α-hydroxylate 16α-[(2)H]-progesterone with 33-44% deuterium retention, indicating stereochemical inversion. We conclude that human CYP17A1 has progesterone 21-hydroxylase activity and human CYP21A2 has progesterone 16α-hydroxylase activity, both of which are enhanced with deuterated substrates. The transition states for C-H bond cleavage in these hydroxylation reactions are either significantly nonlinear and/or asymmetric, and C-H bond breakage is partially rate-limiting for all reactions.
Figures



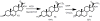
References
-
- Cooper DY, Levin S, Narasimhulu S, Rosenthal O, Estabrook RW. Photochemical action spectrum of the terminal oxidase of mixed function oxidase systems. Science. 1965;147:400–402. - PubMed
-
- White PC, Speiser PW. Congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Endocr Rev. 2000;21:245–291. - PubMed
-
- Therrell BLJ, Berenbaum SA, Manter-Kapanke V, Simmank J, Korman K, Prentice L, Gonzalez J, Gunn S. Results of screening 1.9 million Texas newborns for 21-hydroxylase-deficient congenital adrenal hyperplasia. Pediatrics. 1998;101:583–590. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

